
 |
| May 13, 2010 |
|
During 2008, an estimated 263,871 drug-related ED visits were made by adolescents. Nearly one tenth of these (8.8 percent, or 23,124 visits) involved a suicide attempt. Many of the ED visits involved multiple drugs, with an overall average of 1.8 drugs (including alcohol) per ED visit (2.0 for males; 1.7 for females). Females accounted for nearly three fourths (72.3 percent) of the ED visits for drug-related suicide attempts among adolescents.
Pharmaceuticals were involved in 95.4 percent of ED visits for drug-related suicide attempts among adolescents (Table 1). Alcohol, sometimes combined with other drugs, was involved in about 1 in 10 (11.4 percent) of these visits, while illicit drugs were involved in 8.8 percent of ED visits.5 Anti-anxiety drugs were involved in 26.2 percent of the visits, and almost half of those visits were accounted for by benzodiazepines (12.6 percent). Antidepressants were involved in 23.0 percent of visits, and more than half of those visits were accounted for by selective serotonin reuptake inhibitors (e.g., fluoxetine). A smaller percentage of visits involved narcotic painkillers (e.g., oxycodone) and attention-deficit/hyperactivity disorder (ADHD) stimulants (e.g., methylphenidate) (5.1 and 1.5 percent of visits, respectively). Pain medications containing acetaminophen, ibuprofen, or aspirin as their main ingredient were involved in 25.4, 14.9, and 4.2 percent of ED visits for drug-related suicide attempts, respectively. Other prescription and over-the-counter (OTC) pharmaceuticals involved in suicide attempts were all found at levels less than 5 percent, and usually less than 1 percent (data not shown).
| Drug Category | Estimated Number of ED Visits** |
Percentage of Visits** |
|---|---|---|
| Total ED Visits | 23,124 | 100.0% |
| Major Substances of Abuse | 3,609 | 15.6% |
| Alcohol | 2,633 | 11.4% |
| Illicit Drugs | 2,041 | 8.8% |
| Marijuana | 1,709 | 7.4% |
| Pharmaceuticals | 22,065 | 95.4% |
| Anti-anxiety Drugs (e.g., Anxiolytics,Sedatives, and Hypnotics) | 6,058 | 26.2% |
| Benzodiazepines (e.g., Alprazolam) | 2,915 | 12.6% |
| Antidepressants | 5,312 | 23.0% |
| Selective Serotonin Reuptake Inhibitors | 3,242 | 14.0% |
| Antipsychotics (e.g., Quetiapine) | 1,710 | 7.4% |
| Narcotic Painkillers (e.g., Oxycodone) | 1,181 | 5.1% |
| Central Nervous System Stimulants (e.g., Attention-deficit/Hyperactivity Disorder Drugs) | 346 | 1.5% |
| Acetaminophen Products | 5,863 | 25.4% |
| Ibuprofen Products | 3,453 | 14.9% |
| Aspirin Products | 966 | 4.2% |
| * Other drugs involved in suicide attempts were all found at levels less than 5 percent, and usually less than 1 percent; therefore, data are not shown. ** Because multiple drugs may be involved in each visit, estimates of visits by drug may add to more than the total, and percentages may add to more than 100 percent. Source: 2008 (08/2009 update) SAMHSA Drug Abuse Warning Network (DAWN). |
The drugs involved in ED visits for drug-related suicide attempts varied by gender (Figure 1). For example, antipsychotic drugs (e.g., quetiapine) were indicated in 14.3 percent of visits by males compared with 4.8 percent of visits by females, whereas acetaminophen products were indicated in 28.5 percent of ED visits by females compared with 17.1 percent of visits by males.
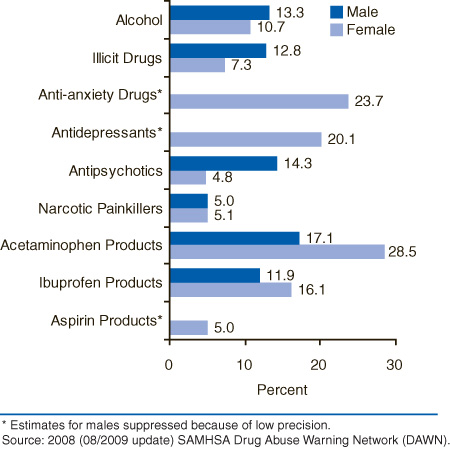 |
| Selected Substances | Male Percent | Female Percent |
|---|---|---|
| Alcohol | 13.3% | 10.7% |
| Illicit Drugs | 12.8% | 7.3% |
| Anti-anxiety Drugs* | 0.0% | 23.7% |
| Antidepressants* | 0.0% | 20.1% |
| Antipsychotics | 14.3% | 4.8% |
| Narcotic Painkillers | 5.0% | 5.1% |
| Acetaminophen Products | 17.1% | 28.5% |
| Ibuprofen Products | 11.9% | 16.1% |
| Aspirin Products* | 0.0% | 5.0% |
| * Estimates for males suppressed because of low precision. Source: 2008 (08/2009 update) SAMHSA Drug Abuse Warning Network (DAWN). |
Overall, more than three fourths (77.0 percent) of the ED visits for drug-related suicide attempts among adolescents ended with evidence of follow-up care. The likelihood of follow-up varied by the types of drugs involved, ranging from a high of 90.2 percent for visits involving prescription antidepressants to a low of 52.4 percent for visits involving ibuprofen products (Figure 2). Follow-up care is defined broadly to include admission to an inpatient unit in the hospital, transfer to another health care facility, or referral to detoxification.
 |
| Selected Type of Drug | Follow-up Percent |
No Follow-up Percent |
|---|---|---|
| Alcohol | 83.1% | 16.9% |
| Illicit Drugs | 80.1% | 19.9% |
| Anti-anxiety Drugs | 75.5% | 24.5% |
| Antidepressants | 90.2% | 9.8% |
| Antipsychotics | 77.9% | 22.1% |
| Narcotic Painkillers | 74.5% | 25.6% |
| Acetaminophen Products | 81.7% | 18.3% |
| Ibuprofen Products | 52.4% | 47.6% |
| Aspirin Products | 71.6% | 28.4% |
| * Percentages for each type of drug involved may not add to 100 percent due to rounding. Source: 2008 (08/2009 update) SAMHSA Drug Abuse Warning Network (DAWN). |
Teen suicide is a major public health problem. One predictor of a future suicide attempt is a previous suicide attempt, and any adolescent who is in an ED because of a suicide attempt should be considered at higher risk for another, possibly fatal, attempt. Thus, EDs afford a unique opportunity to identify at-risk youth and their families and to provide immediate intervention. The findings reported here point to several considerations that may help policymakers, health care professionals, educators, and parents to take advantage of this intervention opportunity.
First, a greater proportion of the ED visits for drug-related suicide attempts were made by adolescent females than males, and the types of drugs involved in the visits varied by gender. Therefore, it may be important to consider these gender differences in managing crises, developing and using brief interventions, and planning aftercare.
Second, an ED visit for a suicide attempt is an opportunity to intervene with the parents/caretakers to educate them about the importance of monitoring the medicines to which the child has access.
Third, it is clear from the findings on ED follow-up that visits entailing less dangerous drugs (e.g., ibuprofen products) receive less follow-up than visits involving prescription drugs such as antidepressants. This may indicate a need to promote awareness among ED professionals and to develop more therapeutic interventions.
| The Drug Abuse Warning Network (DAWN) is a public health surveillance system that monitors drug-related morbidity and mortality. DAWN uses a probability sample of hospitals to produce estimates of drug-related emergency department (ED) visits for the United States and selected metropolitan areas annually. DAWN also produces annual profiles of drug-related deaths reviewed by medical examiners or coroners in selected metropolitan areas and States. Any ED visit related to recent drug use is included in DAWN. All types of drugs—licit and illicit—are covered. Alcohol is included for adults when it occurs with another drug. Alcohol is always reported for minors even if no other drug is present. DAWN's method of classifying drugs was derived from the Multum Lexicon, Copyright 2008, Multum Information Services, Inc. The Multum Licensing Agreement can be found in DAWN annual publications at http://www.multum.com/license.htm. DAWN is one of three major surveys conducted by the Substance Abuse and Mental Health Services Administration's Office of Applied Studies (SAMHSA/OAS). For more information on other OAS surveys, go to http://www.oas.samhsa.gov/. SAMHSA has contracts with Westat (Rockville, MD) and RTI International (Research Triangle Park, NC) to operate the DAWN system and produce publications. For publications and additional information about DAWN, go to http://DAWNinfo.samhsa.gov/. |
| The DAWN Report is published periodically by the Office of Applied Studies, Substance Abuse and Mental Health Services Administration (SAMHSA). All material appearing in this report is in the public domain and may be reproduced or copied without permission from SAMHSA. Additional copies of this report or other reports from the Office of Applied Studies are available online: http://samhsa.gov/data/. Citation of the source is appreciated. For questions about this report, please e-mail: shortreports@samhsa.hhs.gov.
DAWN_001 |
This page was last updated on September 29, 2009. |