Medical Devices
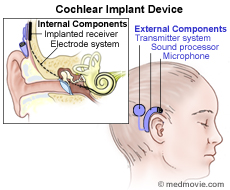
Cochlear Implants
 |
|
| • | View movie of the normal ear, ear with hearing loss, and cochlear implant procedure (Requires Flash Player - Download now) |
Welcome to the Food and Drug Administration (FDA) website on cochlear implants. Cochlear implants are electronic hearing devices. Doctors implant cochlear implants into people with severe to profound hearing loss to produce useful hearing sensations.
The purpose of this website is to describe cochlear implants, link to FDA-approved implants, tell the benefits and risks of cochlear implants, and provide news about cochlear implant recalls and safety issues. You can find information here too on what educators of implant users need to know, what happens before, during and after surgery, and where to report problems.
The FDA regulates manufacturers of cochlear implants. For manufacturers to sell cochlear implants in the United States, they must first show the FDA that their implants are safe and effective. As a matter of policy, FDA does not rate or recommend brands of cochlear implants or medical facilities that implant them.