-

2012
ChemBioChem
The cover picture shows the binding of a PLHSpT derivative, 6q, to the polo-like
kinase 1 (Plk1) polo-box domain (PBD), thereby uncovering a new hydrophobic channel
(magnified upper right), which is absent in the unliganded protein (magnified lower
left). The authors explain how, as a consequence of the additional interaction with
the channel, the peptide binds to the Plk1 PBD with a binding affinity more than
two orders of magnitude higher. The background image of a stained cell nucleus depicts
how this binding results in interference with Plk1 (red dots)-dependent bipolar
spindle formation (green), and this ultimately leads to mitotic block and apoptotic
cell death in cultured cancer cells. See: Peptoid–Peptide Hybrid Ligands
Targeting the Polo Box Domain of Polo-Like Kinase 1k by Fa Liu,
Jung-Eun Park, Wen-Jian Qian, Dan Lim, Andrej Scharow, Thorsten Berg, Michael B.
Yaffe, Kyung S. Lee*, and Terrence R. Burke Jr., in the ChemBioChem ,
2012, 13, 1291-1296.
-
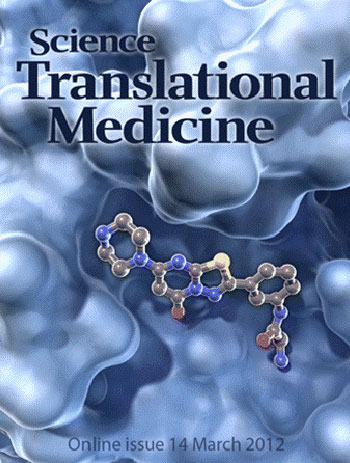
2012
Science Translational Medicine
A Better Fit. An improved anticoagulant drug called RUC-2 (ball and stick structure)
fits snugly into its binding pocket on integrin (blue), a protein found on the surface
of platelets. RUC-2 binds both subunits of integrin, inhibiting the excessive blood
coagulation that can lead to strokes and heart attacks. Unlike similar drugs that
alter integrin's structure when they bind and trigger unwanted immune responses,
RUC-2 does not disturb the configuration of its larger partner. See: Structure-Guided
Design of a High-Affinity Platelet Integrin αIIbβ3 Receptor Antagonist That Disrupts
Mg2+ Binding to the MIDAS by Jieqing Zhu, Won-Seok Choi, Joshua
G. McCoy, Ana Negri, Jianghai Zhu, Sarasija Naini, Jihong Li, Min Shen, Wenwei Huang,
Daniel Bougie, Mark Rasmussen, Richard Aster, Craig J. Thomas, Marta Filizola, Timothy
A. Springer and Barry S. Coller in the Science Translational Medicine,
2012, 4(125), 125ra32. [CREDIT: C. BICKEL/SCIENCE TRANSLATIONAL
MEDICINE].
-

2012
Journal of Organic Chemistry
Beginning with a known 3-oxabicyclo[3.1.0]-hexane scaffold, the relocation of the
fused cyclopropane ring bond and the shifting of the oxygen atom to an alternative
location engendered a new 2-oxabicyclo[3.1.0]hexane template that mimics more closely
the tetrahydrofuran ring of conventional nucleosides. The synthesis of this new
class of locked nucleosides involved a novel approach that required the isocyanate
with a hydroxyl-protected scaffold as a pivotal intermediate that was obtained in
11 steps from a known dihydrofuran precursor. See: Synthesis of Conformationally
North-Locked Pyrimidine Nucleosides Built on an Oxabicyclo[3.1.0]hexane Scaffold
by Olaf R. Ludek and Victor E. Marquez in the Journal of Organic Chemistry,
2012, 77(1), 815-824.
-

2011
Peptide Science
In silico-generated hypothetical interactions of a ring-closing metathesis-macrocylized
peptide bound to the amino terminal SH3 domain of the growth factor receptor bound
protein 2 (Grb2). The complex was derived from the NMR solution structure of the
bound parent peptide, Ac-V-P-P-P-V-P-P-R-R-R-amide (Protein Data Bank: 3GBQ). The
protein surface is shown as electrostatic potential (blue = positive; red = negative).
See: Application of Ring-Closing Metathesis to Grb2 SH3 Domain-Binding Peptides
by Fa Liu, Alessio Giubellino, Philip C. Simister, Wenjian Qian, Michael C. Giano,
Stephan M. Feller, Donald P. Bottaro and Terrence R. Burke Jr. in Peptide Science,
2011, 96 (6), 780–788.
-

2011
Organic Chemistry
Bridging bioinorganic chemistry with asymmetric synthesis: a naturally occurring
metalloprotein is used for the structure-based evolution of chiral auxiliaries that
prove to be effective in the synthesis of Fmoc-L-γ-carboxyglutamic acid. See
Enhanced Stereoselectivity of a Cu(II) Complex Chiral Auxiliary in the Synthesis
of Fmoc-L-γ-carboxyglutamic Acid by Daniel Smith, Glenn Yap,
James Kelley and Joel Schneider in the Journal of Organic Chemistry,
2011, 76 (6), pp 1513–1520.
-
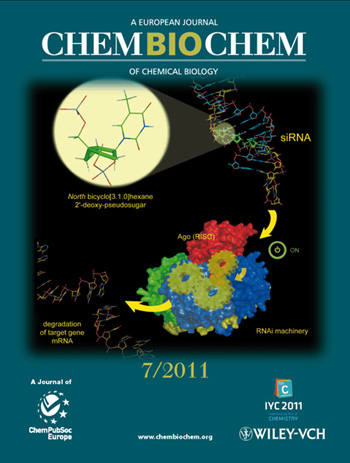
2011
ChemBioChem
The inside cover picture shows how siRNAs modified with North bicyclo[3.1.0]hexane
2'-deoxy-pseudosugars are able to activate the RNA interference machinery. The paper
confirms that the North conformation is critical for RNAi activity. For further
details, see Effect of North Bicyclo[3.1.0]hexane 2'-Deoxypseudosugars on
RNA Interference: A Novel Class of siRNA Modification by Montserrat
Terrazas, Sandra M. Ocampo, José Carlos Perales, Victor E. Marquez and Ramon Eritja
in ChemBioChem 2011, 12 (7), 1056 – 1065.
-
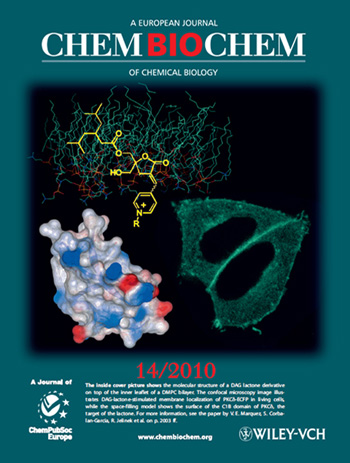
2010
ChemBioChem
The inside cover picture shows the molecular structure of a DAG lactone derivative
on top of the inner leaflet of a DMPC bilayer. The confocal microscopy image illustrates
DAG-lactone-stimulated membrane localization of PKCδ-ECFP in living cells, while
the space-filling model shows the surface of the C1B domain of PKCδ, the target
of the lactone. For more information, see: Membrane-Surface Anchoring of
Charged Diacylglycerol-Lactones Correlates with Biological Activities
by Or Raifman, Sofiya Kolusheva, Said El Kazzouli, Dina M. Sigano, Noemi Kedei,
Nancy E. Lewin, Ruben Lopez-Nicolas, Ana Ortiz-Espin, Juan C. Gomez-Fernandez, Peter
M. Blumberg, Victor E. Marquez, Senena Corbalan-Garcia and Raz Jelinek in ChemBioChem
2010, 11 (14), 2003-2009.
-

2009
ChemMedChem
The picture shows the locked north (blue) and south (red) bicyclo[3.1.0]hexane nucleosides
in the normal pseudorotational cycle, and the corresponding shift to a smaller cycle
(nmax=7°) caused by the insertion of a double bond. The former nucleosides are inactive,
while the flattening of the embedded cyclopentene ring provides active compounds
against HOS cells infected with HIV. See: North- and South-Bicyclo[3.1.0]Hexene
Nucleosides: The Effect of Ring Planarity on Anti-HIV Activity
by Pamela L. Russ, Maria J. Gonzalez-Moa, B. Christie Vu, Dina M. Sigano, James
A. Kelley, Christopher C. Lai, Jeffrey R. Deschamps, Stephen H. Hughes and Victor
E. Marquez in ChemMedChem 2009, 4 (8), 1354 -
1363.
-

2006
Proceedings of the National Academy of Sciences
The lowest energy-binding conformation of an inhibitor bound to the dimeric interface
of HIV-1 integrase core domain. The yellow region represents a unique allosteric
binding site identified by affinity labeling and mass spectrometry and validated
through mutagenesis. This site can provide a potential platform for the rational
design of inhibitors selective for disruption of integrase multimerization. See:
Discovery of a small-molecule HIV-1 integrase inhibitor-binding site
by Laith Q. Al-Mawsawi, Valery Fikkert, Raveendra Dayam, Myriam Witvrouw, Terrence
R. Burke, Jr., Christoph H. Borchers and Nouri Neamati in PNAS 2006,
103 (26), 10080-10085.
-

2005
Journal of Medicinal Chemistry
Interaction of the β-L-enantiomer of a 4'-C-ethynyl-2',3'-dideoxynucleoside analogue
(ent-14-TP) at the active site of HIV reverse transcriptase. See: A 4'-C-Ethynyl-2',3'-Dideoxynucleoside
Analogue Highlights the Role of the 3'-OH in Anti-HIV Active 4'-C-Ethynyl-2'-deoxy
Nucleosides by Maqbool A. Siddiqui, Stephen H. Hughes, Paul L.
Boyer, Hiroaki Mitsuya, Que N. Van, Clifford George, Stefan G. Sarafinanos, and
Victor E. Marquez in the Journal of Medicinal Chemistry 2004,
47, 5041-5048.
-

2004
Nucleic Acids Research
HHAI methyltransferase (blue ribbon) bound to oligonucleotide (strands with bonds
colored yellow and green) containing a pseudorotationally constrained sugar analogue
at the target position (orange bonds with cyan atoms). The south-constrained pseudosugar
is rotated about its flanking phosphodiester bonds, 90° from its initial position
in B-form DNA, but short of a completely flipped position with 180° rotation. Thus,
it is trapped in the middle of the flipping pathway via the major groove side. This
structure provides clues to DNA-protein interactions in a potential transition state.
See: Caught in the act: visualization of an intermediate in the DNA base-flipping
pathway induced by HhaI methyltransferase by John R. Horton, Gary
Ratner, Nilesh K. Banavali, Niu Huang, Yongseok Choi, Martin A. Maier, Victor E.
Marquez, Alexander D. MacKerell Jr and Xiaodong Cheng in Nucleic Acids Res.
2004, 32 (13), 3877–3886.
-
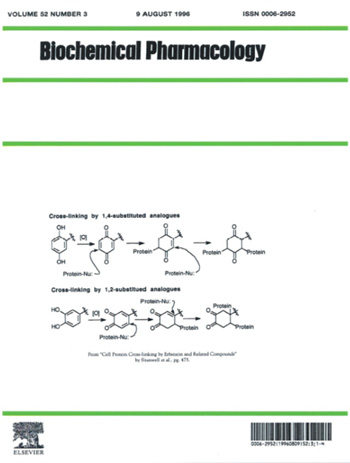
1996
Biochemical Pharmacology
The scheme depicts a possible mechanism of cross-linking by erbstatin and related
analogues. A mechanism of action is proposed which involves initial oxidation to
reactive quinone intermediates that subsequently cross-link protein nucleophiles
via multiple 1,4-Michael-type additions. Similar alkylation of protein by protein-tyrosine
kinase inhibitors, such as herbimycin A, has been invoked. See: Cell protein
cross-linking by erbstatin and related compounds by Caroline Stanwell,
Bin Ye, Stuart H Yuspa and Terrence R Burke Jr. in Biochemical Pharmacology
1996, 52 (3), 475-480.