Digital Press Kit
CDC Responds to Multistate Meningitis Outbreak
The Centers for Disease Control and Prevention (CDC) and the Food and Drug Administration (FDA) are currently coordinating a multistate investigation of fungal infection among patients who received a steroid injection with a potentially contaminated product either into the spinal area or in a joint space, such as a knee, shoulder or ankle.
CDC continues to confirm the presence of the fungus Exserohilum in clinical specimens from people with meningitis. At this time, only one clinical specimen has tested positive for the fungus Aspergillus.
Clinicians should continue to contact patients who have received medicines associated with the following three lots of preservative-free methylprednisolone acetate (80mg/ml) from the New England Compounding Center (NECC) that were recalled on September 26, 2012.
- Methylprednisolone Acetate (PF) 80 mg/ml Injection, Lot #05212012@68, BUD 11/17/2012
- Methylprednisolone Acetate (PF) 80 mg/ml Injection, Lot #06292012@26, BUD 12/26/2012
- Methylprednisolone Acetate (PF) 80 mg/ml Injection, Lot #08102012@51, BUD 2/6/2013
The potentially contaminated injections were given starting May 21, 2012. See updated Clinician Guidance webpage for more information.
CDC's guidance to patients has not changed as a result of the expanded voluntary recall of all NECC products![]() , announced on October 6, 2012.
, announced on October 6, 2012.
Patients who believe they might have received a potentially contaminated medication should contact the physician who performed their procedure to find out if their medication was from one of the three lots.
Patients who received a potentially contaminated medication should seek medical attention if they have any symptoms.
Patients need to remain vigilant for onset of symptoms because fungal infections can be slow to develop. Typically in this outbreak, symptoms have appeared 1 to 4 weeks following injection, but it’s important to know that longer and shorter periods of time between injection and onset of symptoms have been reported. Therefore, patients and physicians need to closely watch for symptoms for at least several months following the injection. For more information, see updated Patient Guidance.
“We are working tirelessly with our state public health partners to track down patients who may have received these medications. If patients are identified soon and put on appropriate antifungal therapy, lives may be saved.”
-Benjamin J. Park, MD, State Response and Surveillance Team Lead for the Multistate Meningitis Outbreak and Chief Epidemiologist with the Mycotic Diseases Branch, US Centers for Disease Control and Prevention
“Given that fungal infections of this kind have never been seen before, the doctors caring for these patients are going to need guidance. CDC has convened the nation’s top clinical fungal experts to work with us in developing diagnostic and treatment guidance for physicians caring for these patients. Patients who are concerned about whether they were exposed to a potentially contaminated product should contact the physician who performed their injection.”
-John Jernigan, MD, MS, Clinical Team Lead of the Multistate Meningitis Outbreak and Director of the Office of Health Associated Infections Prevention Research and Evaluation, Division of Healthcare Quality Promotion, US Centers for Disease Control and Prevention
“Patients deserve to be safe wherever they receive their medical care. This tragic outbreak highlights the need to ensure that all products, materials, and procedures used in healthcare are safe.”
-J. Todd Weber, MD, Incident Manager of the Multistate Meningitis Outbreak and Chief of the Prevention and Response Branch of the Division of Healthcare Quality Promotion, US Centers for Disease Control and Prevention
- CDC, FDA, Massachusetts Department of Public Health Joint Telebriefing Updating Investigation of Meningitis Outbreak Thursday, October 11, 2012 at 3 p.m.
- Media Advisory: CDC, FDA, Massachusetts Department of Public Health Joint Telebriefing Updating Investigation of Meningitis Outbreak, October 11, 2012
- Press Release: Update on Nationwide Meningitis Outbreak, October 5, 2012
- Press briefing transcript: CDC and FDA Joint Telebriefing on Investigation of Meningitis Outbreak, October 4, 2012
- MMWR: Multistate Outbreak of Fungal Infection Associated with Injection of Methylprednisolone Acetate Solution from a Single Compounding Pharmacy — United States, 2012
CDC Resources:
- Multistate Meningitis Outbreak Investigation
- COCA Conference Calls
- CDC Fungal Diseases
- CDC Fungal Meningitis
- Aspergillosis
- Exserohilum
For Clinicians
- Frequently Asked Questions For Clinicians: Multistate Meningitis Outbreak Investigation
- What Should Physicians Be Doing?
Health Alert Network
- CDC Health Alert Network (HAN): Multistate Outbreak of Meningitis and Stroke Associated with Potentially Contaminated Steroid Medication
- CDC Health Alert Network (HAN): Meningitis and Stroke Associated with Potentially Contaminated Product
Guidance
- Interim Treatment Guidance for Central Nervous System and/or Parameningeal Infections Associated with Injection of Potentially Contaminated Steroid Products
- Interim Treatment Options for Septic Arthritis Associated With Injection of Potentially Contaminated Steroid Products
- Interim Guidance for Management of Asymptomatic Persons Exposed to Potentially Contaminated Steroid Products
Case Definitions
Testing
Prophylaxis
For Patients and the Public
- Frequently Asked Questions for Patients
- Healthcare Associated Infections
- National Center for Emerging and Zoonotic Infectious Diseases
Additional Resources:
CDC works 24/7 saving lives and protecting people from health threats to have a more secure nation. Whether these threats are chronic or acute, manmade or natural, human error or deliberate attack, global or domestic, CDC is the U.S. health protection agency.
CDC Media Relations
(404) 639-3286
media@cdc.gov


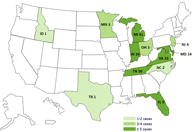
Current Case Count Map
Get email updates
To receive email updates about this site, enter your email address:
Contact Us:
- Centers for Disease Control and Prevention
1600 Clifton Rd
Atlanta, GA 30333 - 800-CDC-INFO
(800-232-4636)
TTY: (888) 232-6348 - Contact CDC-INFO