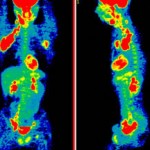
NCI’s Cancer Imaging Program (CIP) is responsible for oversight and funding of oncology imaging research, such as the application of MRI (magnetic resonance imaging) and PET (positron emission tomography) in clinical trials. However, the larger goal of CIP is the development of new imaging techniques and their application to the development of new therapies for the cancer patient.
In phase 0 trials (for imaging agents) the goal is the development of assays that are verified in preclinical models that can be applied to first in-human studies to evaluate relevant biomarkers related to the target of interest. These studies, as would be expected, precede the established phase I through III trials that are required to establish safety and efficacy. These first in human studies are performed on a very small number of eligible trial participants. One of the underlying rationales for phase 0 studies of imaging agents is the intrinsic shortcomings of some animal models, specifically, the transferability of animal model data to humans.
 NCI NewsCenter
NCI NewsCenter NCI Budget Data
NCI Budget Data Visuals Online
Visuals Online NCI Fact Sheets
NCI Fact Sheets Understanding Cancer Series
Understanding Cancer Series
