Transparency
We believe that transparency and data sharing are of fundamental importance to our ability to achieve HHS’s strategic goals of advancing the health and well-being of the United States. HHS’s vast stores of data are a remarkable national resource which can be utilized to help citizens understand what we do and hold us accountable, help the public hold the private sector accountable, increase awareness of health and human services issues, generate insights into how to improve health and well-being, mobilize public and private sector action and innovation to improve performance, and provide the basis for new products and services that can benefit the American people.
The following table describes key audiences for HHS information and the information we seek to make available to them to meet their needs:
Audience | Types of Information and Activities |
|---|---|
| Researchers | Scientific research studies Surveillance, epidemiologic, and risk assessment studies Data files and statistical data Analytical and modeling studies Programmatic guidance “Accountability information” – information on the performance of HHS in the execution of its duties and responsibilities |
| Health, Human Services Practitioners | Scientific research studies Public health surveillance, epidemiologic, and risk assessment studies Data files and statistical data Analytical modeling studies Programmatic guidance Program evaluations Authoritative information targeted to professionals Administrative and regulatory information “Accountability information” – information on the performance of HHS in the execution of its duties and responsibilities |
| Health, Human Services Regulated Industries | Programmatic guidance Administrative and regulatory information Authoritative information targeted to professionals “Accountability information” – information on the performance of HHS in the execution of its duties and responsibilities |
| Business | Authoritative information targeted to consumers, worksites and employers “Accountability information” – information on the performance of HHS in the execution of its duties and responsibilities |
Patients and Clients
| Scientific research studies Public health surveillance, epidemiologic, and risk assessment studies Authoritative information targeted to consumers “Accountability information” – information on the performance of HHS in the execution of its duties and responsibilities |
| Individuals and Families | Authoritative information targeted to consumers “Accountability information” – information on the performance of HHS in the execution of its duties and responsibilities |
| Partners, Grantees and Beneficiaries | Programmatic guidance Administrative and regulatory information “Accountability information” – information on the performance of HHS in the execution of its duties and responsibilities |
| State, Local, and Tribal Governments | Scientific research studies Public health surveillance, epidemiologic, and risk assessment studies Data files and statistical data Analytical modeling studies Programmatic guidance Program evaluations Authoritative information targeted to professionals and consumers “Accountability information” – information on the performance of HHS in the execution of its duties and responsibilities |
| Media | Press releases Fact sheets Scientific research studies Public health surveillance, epidemiologic, and risk assessment studies Analytical and modeling studies Administrative and regulatory information Authoritative information targeted to professionals and consumers “Accountability information” – information on the performance of HHS in the execution of its duties and responsibilities |
| Oversight Groups | Scientific research studies Public health surveillance, epidemiologic, and risk assessment studies Data files and statistical systems Analytical modeling studies Programmatic guidance Program evaluations Authoritative information targeted to professionals and consumers Administrative and regulatory information “Accountability information” – information on the performance of HHS in the execution of its duties and responsibilities |
What follows is a plan that will significantly ramp up how we leverage HHS data to accomplish our stated objectives and serve these audiences. It’s a plan that revolves around four core principles:
- Publish more government information online in ways that are easily accessible and usable
- Develop and disseminate accurate, high quality, and timely information
- Foster the public’s use of the information we provide
- Advance a culture of data sharing at HHS
3.1 HHS Data Currently Available for Download
HHS has already posted 117 data sets and tools on Data.Gov since its debut in May 2009. Check out the inventory of them on our Open Government website www.hhs.gov/open or at www.data.gov. These data sets and tools include:
- Hospital-by-hospital quality performance statistics compiled by the Centers for Medicare and Medicaid Services (CMS) and which can help inform consumer choices regarding where to get care. Also available: similar information on nursing homes, dialysis facilities, home health agencies
- A regularly updated data set representing all technologies available for licensing from the National Institutes of Health (NIH) and the Food and Drug Administration (FDA), helpful to entrepreneurs and companies looking to drive innovation
- A household cleaning products data set that links over 4,000 consumer brands to health effects as submitted by manufacturers and which allows scientists and consumers to research products based on chemical ingredients
- Detailed summaries of Medicare expenditures on physician services, which allow the public to understand patterns of Medicare spending and analyze the types of services being delivered to address the health needs of the Medicare population. (This data set was first added on January 22 as one of HHS’s new “high value” data sets under the Open Government Directive)
- CDC WONDER, which provides access to online databases, reports, references, and links to external data systems containing a wide range of highly valuable public health information. Data sets that can be queried online from WONDER are continually updated and include data sets related to Acquired Immune Deficiency Syndrome (from 1981), births (starting in 1995), cancer registry statistics (beginning in 1999), mortality data (1979-2006), population estimates (beginning in 1970), sexually transmitted disease (STD) morbidity (1984-2008), tuberculosis case reports (1993-2007), and vaccine adverse events reports (1990-2010). CDC WONDER currently hosts 42 searchable online databases, holding over 200 gigabytes of data. In addition, CDC WONDER allows access to reports, statistics, standard reference tables, and historical guidelines. The wonder.cdc.gov website services over 47 million requests a year. CDC WONDER is widely used by public health programs, researchers, and schools of public health curricula. CDC WONDER has over 1000 citations as a data source for scientific papers and articles
- A downloadable data set which lists all NIH-funded research grants, contracts, and intramural projects from 2005-2009, abstracts for these projects, citation information for publications that acknowledged support from any of these projects, and patents reported by investigators funded by these NIH projects. This data set was published on January 22 as one of HHS’s new “high value” data sets under the Open Government Directive. NIH had received many public requests for this information to be made available in downloadable form. Patient advocates are enthusiastic about this dataset because it makes information available on grants and publications that they had been amassing manually. The biomedical research community is interested in analyzing the data to find collaborators, develop literature bases, and analyze trends in biomedical research. Policy makers and evaluators are interested in analyzing research funding, programs supported, and the results of those programs. Venture capitalists and pharmaceutical companies are interested in analyzing the files to understand the latest trends in federally-funded research
- And much more
New High-Value Data Sets and Tools
“High value” is defined by the Open Government Directive as information which can be used to increase agency accountability and responsiveness, improve public knowledge of the agency and its operations, further the core mission of the agency, create economic opportunity, or respond to need and demand as identified through public consultation. As described earlier, the Chief Information Officer Council and Data Council at HHS have executed an initial review of the current inventory of HHS data to see what additional high value data can be released. A directory of major HHS data and statistical systems is available at http://www.hhs-stat.net/scripts/meta_des_new_17.cfm and is continually updated, and an inventory of Office of Management and Budget-approved data collection projects is available at http://www.reginfo.gov/public/do/PRAMain.
As a result of this initial portfolio review, HHS has decided to release 14 new high-value data sets and three new high-value tools on Data.gov by the end of 2010. Of the 14 new high-value data sets, 12 have never previously been available to the public in any form -- 10 data sets from the Centers for Medicare and Medicaid Services (CMS) alone. (The other two data sets are either online for the first time or downloadable in open format for the first time). The three tools – the CMS Dashboard, FDA-TRACK, and the Office of the National Coordinator for Health Information Technology’s Dashboard – will be brand new.
In addition to these 17 new data sets and tools, HHS will be posting more than 40 existing data sets and tools to Data.gov by the end of 2010 – data and tools which are already available online in some form but will be updated with respect to their content and formally registered on Data.gov
We present key highlights, below, with a particular focus on two divisions of HHS that are emerging as leaders of the next generation of transparency at HHS: the Centers for Medicare and Medicaid Services (CMS) and the Food and Drug Administration (FDA).
Centers for Medicare and Medicaid Services (CMS)
We are delighted to announce that CMS is embarking upon a transparency program of unprecedented scope and energy – a development of great importance to HHS and to the transformation of health care. Information is the lifeblood of health care improvement work. Without information, it is impossible to truly understand the current state of health care costs and quality and determine how to improve it. CMS is uniquely positioned to provide such information – and thereby providing transparency both into its own operations and also into American health care itself.
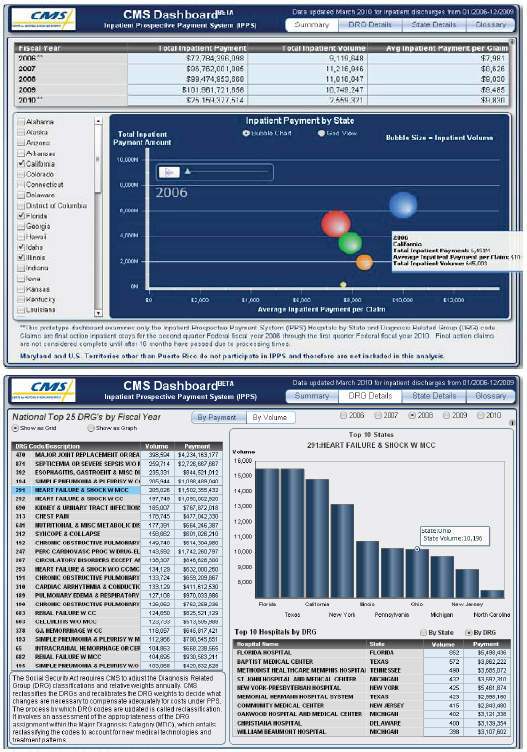
1. The CMS Dashboard. Launched in “beta” mode on CMS’s website on April 6, the CMS Dashboard (http://www.cms.gov/Dashboard/) is one of HHS’s official “Open Government Flagship Initiatives”
- The Dashboard is an exciting new web application which allows the public to visualize and analyze Medicare spending with unprecedented ease and clarity – beginning with inpatient hospital spending
- The initial version of the Dashboard allows users to track and graph Medicare spending on inpatient hospital services by state, by the top 25 “diagnosis-related groups” (DRG), and by top 10 hospitals for each state and diagnosis-related group over time. Users can also see how much Medicare is spending to support important public policy goals, such as the provision of medical education, additional funding to hospitals that take a disproportionate share of low-income patients, additional payments to rural hospitals, additional funding to hospitals who take cases that incur extraordinarily high costs of treatment, and adjustments for the underlying costs in different geographic regions.
- While much of this data had previously been available in some form to the public, scattered across hard copy and electronic publications, analysis of the data required retrieval of it from disparate places and having your own computer programmer and in-house Medicare expert to pull it together – barriers which the Dashboard now eliminates
- Furthermore, the Dashboard makes Medicare inpatient hospital spending data available to the public in a much more timely way than ever before. Data for a given quarter is published less than three months after the end of that quarter, vs. a previous publication lag time of up to 18 months
- Finally, the beta version of the Dashboard is just the beginning. After moving the Dashboard to full production mode, we plan to evolve and grow the Dashboard on an ongoing basis, adding more and more data and functionality – beginning with the next major release of the Dashboard in the fall of 2010
- The Dashboard will provide the public, researchers, policymakers, health care providers, and others with key information on the status of and emerging trends in Medicare service utilization and spending – insights which will help power better-targeted, more timely work to improve our health care system
Sample Screenshots from the CMS Dashboard
2. Creation of 9 Medicare claim “basic files,” including one for each major category of health care service, to be released from September to December 2010 for free public download on Data.gov. These files will contain a limited number of variables and be de-identified and configured through a rigorous process, in close consultation with privacy experts, re-identification experts, researchers, and key stakeholders, to ensure the protection of beneficiary privacy and confidentiality
- As a by-product of administering Medicare, CMS maintains a comprehensive database of claims for all traditional Medicare beneficiaries (about 80% of the Medicare population) – claims from different settings and types of care, including (1) inpatient hospital, (2) outpatient hospital, (3) skilled nursing facilities, (4) home health, (5) hospice, (6) carrier (physicians and suppliers), (7) durable medical equipment, and (8) prescription drugs
- To date, access to these claims has been very restricted
- CMS is initiating a project to improve access to its data by creating “basic files” from claims data for public use
- The primary product will be a set of “basic files” containing 2008 claims data for a randomly selected 5% sample of the Medicare population (about 2.4 million beneficiaries). There will be a separate file for each of the eight claim types noted above. CMS also plans to create an “enrollment file” describing the number and the demographics of the beneficiaries in the 5% sample to support the calculation of population-based rates and proportions
- The files will be carefully stripped of information and include only a limited number of variables– hence the term “basic files.” This work will be executed through a rigorous and intensive process, consulting closely with privacy experts, re-identification experts, researchers, and key stakeholders, to ensure that beneficiary confidentiality is entirely protected. As a key note, each of these 9 files will be “stand-alone” files, published without identifiers that link claims across files to a given beneficiary -- to ensure that a beneficiary cannot be re-identified through their pattern of claims. The de-identification protocol applied to these files will also be such that claims will not be able to be linked to individual providers
- We are planning to make these 9 files available to the public, downloadable at no charge, through Data.gov. The first of the claim “basic files” will be released in September 2010, and all 9 of them will be published by December 2010
- This will represent the first time these kinds of data have been made available to the public
- These files will allow users to study health care services provided through the Medicare program in ways never possible before. The effort is intended to enhance Medicare program transparency and can provide a vital spark for new research and innovation, ultimately leading to improved outcomes for Medicare beneficiaries and the Medicare program.
3. Significantly improved user interface and analytical tool for the public to use to access CMS’s existing COMPARE data on health care provider quality performance – launched with the roullout of this Open Government Plan at data.medicare.gov
- As mentioned previously, one of the most potent and useful data sets CMS has published on Data.gov is detailed quality performance data for individual hospitals, nursing homes, home health agencies, and dialysis providers across the country – a data set called “COMPARE”
- This data can help consumers make better care decisions. Providers are using COMPARE data as an important tool to help them guide their quality improvement efforts, with measurable and discernible results. The COMPARE data set helps researchers gain new insights into care delivery, and aids policymakers in their decisionmaking
- While quite powerful, this data set has been difficult for users to utilize and manipulate in the past, hampering its impact
- At data.medicare.gov, as of the rollout of this Open Government Plan, CMS has now launched a new user interface which the public can use to not only view the data much more easily, but also to customize that view by providing sort, search, and filter capabilities. In addition, users will be able to socialize and share the data through social media networking sites such as Facebook, Twitter, etc.
4. Medicare Part B National Summary File – now downloadable at no charge
- This file provides a detailed breakdown of the volume of physician services delivered to Medicare beneficiaries and how much Medicare paid for those services – by type of physician procedure. It can be used to look at trends in types of services delivered over time or to better understand the magnitude of the health needs of the Medicare population
- This much-in-demand file was previously available for a fee of $100 per year and only on CD ROM
- On January 22, as part of the new high value data sets HHS delivered to the public as per the Open Government Directive, CMS made the 2008 Part B National Summary File available downloadable from Data.gov for free. Files from 2000-2007 are now also available for download from the CMS website for free
5. Medicaid State Plan Documents – to be put online on CMS’s website by the end of 2010
- CMS has initiated a multi-stage project to place the detailed formal documents describing each state’s Medicaid Plan (each of which is unique) on the CMS website, much as the documents describing the Children’s Health Insurance Program State Plans are currently on the CMS website
- Through the Medicaid program, states and the federal government fund a wide range of health services for low income families, pregnant women, children, the elderly, and the disabled
- The Medicaid State Plans are the legally binding documents that serve as contracts between the federal and state government. Each State Plan outlines in detail the nature of that state’s particular Medicaid program, including benefits provided, populations covered and extensive details on the program’s structure, financing and management. They are each accompanied by a stream of amendments that modify the program on an ongoing basis
- Putting the State Plans and their associated amendments online will promote accountability and provide valuable information for citizens about what their government is doing
- The process of compiling all plans and amendments, ensuring that they are complete and accurate, validating them with the states, and putting them online on the CMS website in a standard format should be completed by the end of 2010.
6. New community-level indicators of health care cost, quality, and utilization, to be supplied by CMS to the new HHS Community Health Data Initiative by the end of 2010
- As a key part of HHS’s new Community Health Data Initiative – another one of HHS’s “Open Government Flagship Initiatives” – CMS will be contributing an exciting new set of metrics that describe community-level Medicare prevalence of disease, quality, costs, and utilization of services across the country at a national, state, regional, and potentially county level
- These community indicator metrics, derived from Medicare data, will either be brand new to the world or at a finer level of precision than has previously been available to the public from other sources
- Combined with other HHS data on the health and health care performance of communities, this vital new CMS data can be used by a wide variety of stakeholders to gain new insights into local health and health care performance and to develop new tools, strategies, and programs to improve the value of health care and health performance in local regions
- For more on the Community Health Data Initiative, see the Flagship Initiatives section
Food and Drug Administration (FDA)
Like CMS, FDA is engaged in an unprecedented push to become more transparent. Two of FDA’s initiatives are highlighted here as official HHS “Open Government Flagship Initiatives” -- the FDA Transparency Initiative and FDA-TRACK.
1. FDA Transparency Initiative
FDA Commissioner Dr. Margaret Hamburg launched the agency’s Transparency Initiative in June 2009. Over the years, some stakeholders have complained about FDA’s lack of transparency. The agency has been referred to by some as a “black box” that makes important decisions without explaining them. The objective of the FDA Transparency Initiative is to render FDA much more transparent and open to the American public. Increasing FDA’s openness will help the agency more effectively implement its mission to promote and protect the public health by providing the public with useful, user-friendly information about agency activities and decision-making.
Commissioner Hamburg formed an internal task force representing key leaders of FDA to oversee the initiative. Commissioner Hamburg asked Dr. Joshua Sharfstein, the Principal Deputy Commissioner of the FDA, to chair FDA’s internal task force, whose members include five of the Agency’s Center Directors, the Chief Counsel, the Associate Commissioner for Regulatory Affairs, and the Chief Scientist. The Task Force was charged with soliciting public input on ways the agency can improve transparency and making recommendations to Commissioner Hamburg for ways the agency can operate more transparently to benefit the public health.
Over the last eight months, the Task Force has held two public meetings, launched an online blog (accessible at http://fdatransparencyblog.fda.gov/), opened a docket, and held listening sessions with representatives of regulated industry.
At the first public meeting, the Task Force solicited comments on how the Agency could improve transparency overall. Thirty five individuals provided comments during the meeting and 335 people attended in person or watched the live webcast of the eight hour session.
At the second public meeting, the Task Force solicited comments on three specific issues related to transparency at the agency: (1) early communication about emerging safety issues concerning FDA-regulated products, (2) disclosure of information about product applications that are abandoned (no work is being done or will be undertaken to have the application approved) or withdrawn by the applicant before approval, and (3) communication of agency decisions about pending product applications. Sixteen individuals participated in the groups convened to discuss each issue as well as during the open public session.; One hundred seventy four people attended the meeting in person or watched the live webcast.
The online blog and the docket have received over 1,380 comments. The blog has offered an opportunity for exchange about specific ideas for transparency at the agency.
The Task Force is also collecting information on how to improve FDA’s transparency to regulated industry. It held three listening sessions with members of regulated industry in January 2010 and have made available the transcripts and summaries of those listening sessions (accessible at http://www.fda.gov/AboutFDA/WhatWeDo/FDATransparencyTaskForce/default.htm).
The Task Force is proceeding with the Transparency Initiative in three phases:
Phase 1: FDA Basics. The first phase is intended to provide the public with basic information about FDA and how the agency does its work. In early January 2010, FDA launched a web-based resource called FDA Basics (accessible at http://www.fda.gov/fdabasics). The launch involved a media call and webinar with bloggers on FDA issues. This resource now includes (1) 115 questions and answers about FDA and the products that the agency regulates, (2) eight short videos that explain various agency activities, and (3) conversations with nine agency officials about the work of their offices.
The initial content for FDA Basics was based on questions and comments the agency frequently receives from the public. FDA will continue to add user-friendly information to the site.
In addition, visitors to FDA Basics can rate the helpfulness of the information provided and suggest additional questions for inclusion in FDA Basics. The agency has received over 2,700 comments from the public since the launch of the FDA Basics resource. Feedback provided by the public is used to update the resource.
Each month, senior officials from FDA product centers and offices host 30 minute online sessions about a specific topic and answer questions from the public about that topic. Each of these sessions is announced on the FDA web site, the online blog, and promoted to other stakeholders FDA employees identify.
The FDA Basics webinar series was launched in February with a webinar on “Access to Investigational Drugs,” hosted by FDA’s Office of Special Health Issues. An audio replay and copy of the PowerPoint slides from the session are available on the FDA Basics web site.
Early reaction to FDA Basics has been positive. One blogger wrote, “[t]he initiative can go a long way toward educating the public about what FDA does—and how—and also provide industry with real-time answers to their daily challenges, ultimately improving product quality and patient safety.” Another blogger wrote,“ [i]t is really well put together, clear and works quite well... The site is not only supportive of transparency, but is highly instructive and educational.”
Phase 2: Public disclosure. The second phase of the FDA Transparency Initiative relates to FDA’s proactive disclosure of information the agency has in its possession, and how to make information about agency activities and decision-making more transparent, useful, and understandable to the public, while appropriately protecting confidential information.
Phase 3: Transparency to regulated industry. The third phase will address ways that FDA can become more transparent to regulated industry, in order to foster a more effective and efficient regulatory process. FDA is now seeking public input through the Federal Register Notice process, and draft recommendations from this phase are expected in the spring of 2010.
2. FDA-TRACK
In addition to the FDA Transparency Initiative, the other FDA project to be designated an official HHS “Open Government Flagship Initiative” is FDA-TRACK, the FDA’s new agency-wide program performance management system. When fully implemented, FDA-TRACK will monitor over 90 FDA program offices through key performance measures that will be gathered on a monthly basis. Each quarter, the FDA-TRACK team will analyze monthly performance data, with senior managers presenting these data to FDA senior leadership.
Very importantly, the public will be able to track the agency’s progress through the FDA-TRACK website – which will debut in “beta” mode on April 7, with most of FDA’s offices represented.
Consistent with the principles of open government, FDA-TRACK adheres to values that comprise its name – Transparency, Results, Accountability, Credibility and Knowledge-sharing.
Transparency – The FDA-TRACK website (fda.gov/fdatrack) enables all interested external and internal visitors to view performance data at the program office level and gain a better understanding of the breadth of FDA’s core responsibilities, as well as see progress on important projects and programs. When fully implemented, the website will include data for over 300 performance measures and 80 key projects at the FDA. The website will be updated regularly so viewers can monitor the accomplishments and trends for each program area. Complementing the work of the Transparency Task Force, FDA-TRACK exemplifies FDA’s commitment to expanding public access to important information.
Results –FDA-TRACK highlights performance measures with relevance to the agency’s public health mission. These include the timeliness of reviews, the number of high risk inspections completed, and the completion of key research projects. Over time, FDA intends to include more measures that reflect public health outcomes across a broad range of agency activities.
Accountability – Developing, tracking and reporting performance measures will improve the agency’s accountability to the public. In addition, internal discussions between FDA’s senior leaders and program office senior management are conducted each quarter to promote sharing of ideas and hold each FDA office accountable for its priorities, plans and results.
Credibility – Sharing information about FDA performance is important for the agency’s credibility. The FDA-TRACK website will provide an unprecedented look into how FDA does its work. The site also allows visitors to submit comments on both the general features of the program as well as specific performance measures. Website hits and feedback will be monitored on a daily basis by the FDA-TRACK team, and suggestions will be considered as part of our continuous improvement efforts. A recent suggestion from early FDA-TRACK work led to the development of FDA-TRACK Dictionaries that provide a plain language explanation for each of the performance measures reported.
Knowledge-Sharing – FDA recognizes that we can improve our operational effectiveness through better collaboration and sharing of ideas. The implementation of FDA-TRACK will enable us to identify common issues and interdependencies among our program offices. As a result, program offices are working with one another as well as reaching out to other agencies to collaborate in achieving their performance objectives.
FDA-TRACK is a federal level adaptation of successful performance management programs from the state and local level. FDA’s Office of Planning organizes and coordinates the FDA-TRACK process with all of the over 90 program offices across the nine FDA Centers and major offices.
Each of the program offices is responsible for collecting and providing monthly data in preparation for reporting performance via the FDA-TRACK dashboard (see Chart 1). The dashboards are presented to senior leadership through quarterly briefings. The data are then posted to the FDA-TRACK website.
Chart 1: Illustrative - FDA-TRACK Dashboard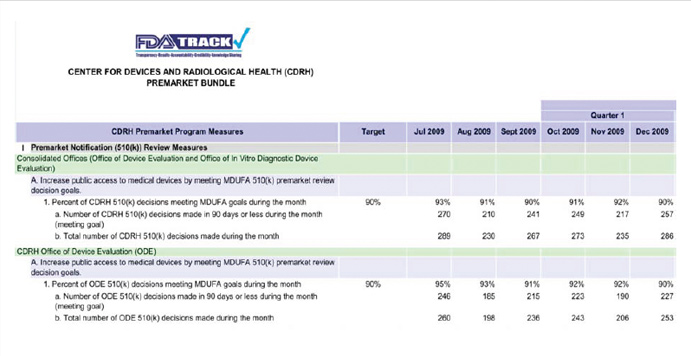
FDA-TRACK publicly reports performance indicators and related data in four categories:
Common MeasuresFDA-wide measures that are applicable to each of the over 90 program offices and may focus on the agency’s most recent priorities. Example: increase the total number of employees who are trained in the Incident Command System (ICS), which helps the agency respond to emergencies.
Key Center Director MeasuresCenter-specific measures that are applicable to each Center and are central to the Center’s priorities and strategic goals. Example: increase the FDA’s technical guidance by increasing the number of FDA technical publications, which enables the Center to better communicate with industry and consumers.
Program Measures - Program office-specific measures that are applicable to the office and reflect work important to the public and FDA’s mission. Example: increase the percentage of 510(k) (or Class II medical devices) decisions made on time during the month (see Chart 2).
Chart 2: Percentage of 510(k) Decisions made on Time
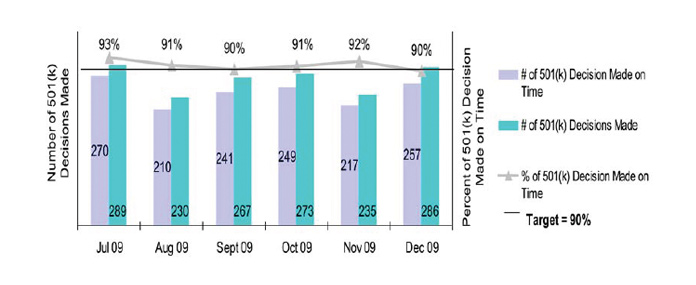
Key Projects - Program office-specific projects that are applicable to the office and important to the mission and objectives of the office. Performance for Key Projects is measured through achievement of the stated milestones within the project’s plan. Example: the development of a new risk-based approach for evaluating safety, effectiveness, and quality of new animal drugs.
The FDA’s senior leaders are committed to making FDA-TRACK successful and sustainable. To accomplish this goal, FDA anticipates continuing to improve this program so it becomes an everyday essential management tool for all program offices. Planned improvements as FDA-TRACK matures include:
- Alignment of FDA-TRACK measures to the annual agency performance measures such as those required by the Government Performance and Results Act of 1993
- Alignment of FDA-TRACK measures to individual employee performance plans
- Improvements to FDA-TRACK data management and reporting software
- Improvements to measures based on public input and experience so that measures can be more closely tied to the public health mission of the agency
- Implementation of improvements to FDA-TRACK performance data analysis to enable better predictive outcomes and other quantitative data-based decision making
FDA is working to make FDA-TRACK a model for open government at the federal level.
3. Other FDA Transparency Actions
In addition to these initiatives, FDA is planning to post new summary aggregate data resulting from the new Reportable Food Registry, beginning in Q4 FY 2010. The FDA Reportable Food Registry contains information about foods for which there is a reasonable probability that the article of food will cause serious adverse health consequences or death. Regulated industry submits reportable food reports to FDA for possible inclusion in the Registry via an electronic portal. Federal, state, and local government officials may also voluntarily use the electronic portal to report information that may come to them about reportable foods. FDA plans to post reports summarizing certain aggregate data from the Registry online. In Q4 FY 2010, FDA also plans to enable the FDA Recalls website to provide drug, device, and food recall data in XML format to empower users to download, reuse or mash-up recall information.
Other Transparency Actions at HHS
In addition to the ambitious agendas being pursued by CMS and FDA, other divisions at HHS are pursuing notable new actions with respect to transparency as well:
- The Administration on Aging (AoA) will be posting a raw data set from the Annual National Survey of Older Americans Act Participants in Q4 FY 2010. This data has never been posted online in a downloadable format or otherwise been made available to the public as a data set.
- The Centers for Disease Control and Prevention (CDC) will be posting a brand new data set -- BioSense Condition-Specific Data, based on reports to the CDC’s BioSense system from participating electronic health records systems (for example, statistics on dental-related emergencies that show up at hospitals).
- The Office of the National Coordinator for Health Information Technology (ONC) is in the process of developing a comprehensive performance measurement system and corresponding performance dashboard (ONC Dashboard) for its activities and programs. This Dashboard will assist ONC in monitoring its many programs and grantees, facilitate assessment of progress toward its goals, and position ONC to serve as a leading example of Open Government. In creating the ONC Dashboard, ONC will use information generated by its programs, its internal processes, its formal evaluations and other data collection efforts as needed. Given the wide range of measures to be collected, the Dashboard will have broad capabilities to support different stakeholders’ need for information. ONC’s performance planning is projected to be complete by late spring, with the beta version of the ONC Dashboard launched for public view before the end of 2010.
Data Set/Tool | Release Date |
|---|---|
CMS Dashboard | Released April 6, 2010 |
9 Medicare claim "basic files” | First file released in September 2010 |
Last file released in December 2010 | |
New user interface/analytical tool for COMPARE data at data.medicare.gov | Released April 7, 2010 |
Medicaid State Plan documents | 4Q 2010 |
New Medicare community-level indicators of health care cost, quality, and utilization to be supplied to the Community Health Data Initiative | 4Q 2010 |
FDA-TRACK | Released April 7, 2010 |
Summary data from the FDA's Reportable Food Registry | 3Q 2010 |
FDA recall data downloadable in XML format | 3Q 2010 |
Administration on Aging Annual National Survey of Older Americans Act -- raw data set | 3Q 2010 |
Centers for Disease Control and Prevention BioSense Condition-Specific Data | 3Q 2010 |
Office of the National Coordinator for Health Information Technology Dashboard | 4Q 2010 |
New HHS Process for Ongoing Data Prioritization, Release and Monitoring
Equal in importance to the exciting new data sets and tools we are releasing is the new process which we will be implementing at HHS in 2010 for the identification, prioritization, publication, and monitoring of data releases. Since the debut of our initial Open Government Plan on April 7, we have launched this process, which will adhere to the following key principles:
- HHS will continually review current inventories and portfolios of data sets and tools and identify high value data sets and tools for electronic dissemination in support of Transparency and Open Government goals. Reviews are anticipated on a quarterly basis.
- At the planning stage for any new data development endeavor or major revision of existing data development efforts, and throughout the life of the project, HHS will identify opportunities for transparency, data sharing and dissemination though electronic posting of datasets. These dissemination activities will become part of the project or system plan for the data project. Since the debut of our initial Open Government Plan on April 7, we have implemented this guidance as part of our HHS Enterprise Architecture program and incorporated it into the ongoing monitoring of IT projects and investments that are accomplished using HHS’s IT investment life cycle planning and execution processes. As opportunities for data sharing and dissemination are identified, they will be evaluated and adjudicated by the HHS Data Council.
- HHS will protect the confidentiality of individually identifiable information in any public data releases, including publication of data sets on the web. Recognizing that the risks of disclosure of personal identifiable information mount as more and diverse data sets are released into the public domain, HHS will lead and support ongoing research into strategies to protect confidentiality while maintaining public access to data. As new approaches evolve, HHS will incorporate them into its data release policies.
- HHS will consider specific audiences, market segments and communication objectives in its data development and dissemination programs.
- HHS will undertake efforts to foster the use and utility of the data it disseminates publicly through the web and seek feedback from data users.
- HHS will ensure the quality, objectivity, integrity and utility of the substantive data it disseminates through compliance with HHS and Agency Information Quality Guidelines. http://aspe.hhs.gov/infoquality
- HHS will employ feedback, outreach and evaluation mechanisms regarding the value and utility of the information we disseminate and use that information to enhance and improve our data products and services in a continuous quality improvement cycle.
Our updated data sharing process will be enmeshed into the department’s core strategic planning, budgeting, and information technology planning activity in 2010. Key points:
- HHS will embed data experts on the strategic planning and management teams for each of the Secretary’s strategic initiatives and cross-departmental priorities. The role of the data experts will be to 1) provide each strategic planning team with knowledge of current HHS data and information relating to the specific initiative, 2) assess data needs, gaps and opportunities, 3) develop plans and recommendations regarding the evaluation and performance information that will be used to implement and assess progress on the initiative, and 4) proactively identify and actively promote ways of sharing existing and new data with the public and key audiences in ways that adhere to transparency principles and advance the initiative. HHS’s Data Council will identify one or more experts for each initiative who will bring not only subject matter knowledge to the team but will also serve as a resource regarding available HHS data systems which may support the initiative as well as principles of performance measures, evaluation and information quality. The data experts themselves will convene on a regular basis as a cross-HHS team to coordinate their work and share best practices across the department. Plans and recommendations emerging from each initiative will be employed by the HHS Data Council in its overall data planning and investment priorities.
- Data production and sharing opportunities proactively targeted by each Secretarial initiative will be added to an ongoing stream of new data sets identified for publication on Data.gov through regular portfolio reviews conducted by each HHS agency and office and coordinated by the Data Council and CIO Council. The Data Council will ensure that data releases are screened for quality, privacy, security, and confidentiality risks before release. Data releases will be prioritized based on alignment with HHS’s strategic goals.
- At the planning stage for any new data development endeavor or major revision of existing data development efforts, and throughout the life of the project, HHS will proactively identify opportunities for transparency, data sharing and dissemination through electronic posting of datasets on Data.gov. These dissemination activities will become part of the project or system plan for the data project. This new aspect of data and systems planning will be incorporated into the core management processes, templates and criteria utilized by HHS’s information technology, investment review, and budgeting processes.
- Freedom of Information Act (FOIA) requests are one of the most reliable ways to know what information the public wants. HHS will establish governance within its FOIA operations to promote the proactive publishing of information and include FOIA officers across the department in transparency and data sharing planning activities. This approach will not only improve public access to information but could also help reduce the FOIA request backlog.
- Records Management – the Open Government Directive requires that if an agency keeps records in an electronic format (including those available online), it should develop a plan for the timely publication of the underlying data in an open, easily-discoverable format, and “as granular as possible." HHS programs will analyze which records can be appropriately published and will develop a method to satisfy this Open Government requirement as we move forward with advancing the records management program. This activity will also assist in reducing the FOIA backlog, since some of the requests for information that HHS receives from the public are related to information that is contained in departmental records. HHS is engaged in an analysis of the current process for managing records and extending that process to the future of records management as a largely electronic process. This analysis will include an evaluation of which HHS electronic records may be the best candidates for publication to and review by the public.
- The Data Council will continue to oversee and manage the process of releasing information assembled via all of the sources above on our Open Government website, Data.gov and other mechanisms.
- A key principle underlying our Open Government work is promoting transparency while maintaining confidentiality. This will require an ongoing effort involving those developing the data and data users and is made all the more challenging by the increasing amount of data from a wide variety of sources that conceivably could be linked to confidential data. The linkage could yield valuable insights but could also lead to the disclosure of confidential information. This challenge is shared by agencies across the government. HHS will work with other agencies to develop confidentiality safeguards for data submitted to Data.gov that will increase the amount and scope of data disseminated while developing ways of preserving the confidentiality of the data's sources, whether individuals or businesses.
- HHS will engage in a proactive new program of monitoring, stimulating, and incorporating innovative and beneficial uses of our data.
- Beginning 3Q 2010, on our Open Government website and through systematic dialogue with key stakeholder groups (overseen by our Data Council), we will solicit examples of how our data has been used to generate benefit. We will seek to compile at least 30 such examples (insights, applications, visualizations, etc.) by the end of 2010 for publication on our Open Government website. This will help us shape our future data release strategy on an ongoing basis.
- We will establish an online forum in 3Q 2010 on our Open Government website that facilitates public discussion of barriers to innovation using our data (e.g., data format, lack of metadata, etc.). Through this forum and other channels, we will seek to gather a list of at least 10 such barriers for publication and discussion of next steps on our Open Government website by the end of 2010.
- Leveraging the new “challenges and prizes” platform to be made available across the government, we will initiate an “HHS Apps Challenge” – a public competition for the most innovative and beneficial applications built utilizing our data. Since the debut of our initial Open Government Plan, we have already executed two such challenges and launched a third:
- A competition for best visualization of community health data as part of the Sunlight Foundation’s Design for America competition -- see http://sunlightlabs.com/blog/2010/design-america-winners/ for results, announced at the end of May
- A challenge to innovators to develop applications using HHS’s community health data for debut at a Community Health Data Forum jointly hosted by HHS and the Institute of Medicine on June 2 – see http://www.hhs.gov/open/datasets/initiative_launch.html to view a webcast of the amazing results, and read more about the Community Health Data Initiative below.
- As part of this Community Health Data Initiative, HHS has collaborated with Health 2.0, Sunlight Foundation, and others to launch a third challenge, the Health 2.0 2010 Developer Challenge (www.health2challenge.org), with resulting applications to be showcased at the Health 2.0 conference in San Francisco in October
- Through the Community Health Data Initiative (a core “Open Government Flagship Initiative,” described in that section of our Open Government Plan), we are pioneering an approach that seeks both to (1) make key HHS data available and easy to access by the public and (2) proactively encourage a growing array of innovators from the worlds of technology, business, academia, public health, and health care to engage with the data and turn it into applications that create significant and growing public benefit
- The Initiative embodies the philosophy of “open data” and data sharing that is at the heart of our Open Government Plan. The core principle is that the government can help trigger enormous public good by implementing the idea of “government as platform,” supplying ever greater amounts of high quality, free government data to the public and marketing the availability of this data – which can then be turned into useful insights, applications, products, and services by private and public sector innovators from across the country, working with engaged consumers, providers, civic leaders, employers, researchers, and others
- The creation of a public-private “ecosystem” of data supply and use is the central objective of the initiative. We are proactively marketing HHS data on community health care, health, and health determinant indicators to innovators who can turn it into applications and programs that benefit the public. We are evangelizing energetically on behalf of the Initiative at conferences and other public forums and on the web, seeking to get many organizations and individuals outside HHS interested in participating in the Initiative’s ecosystem. We are working closely with organizations such as the Institute of Medicine and Health 2.0 to hammer out how we can all facilitate the ecosystem’s growth and development
- As showcased at the June 2 Community Health Data Forum, co-hosted by the Institute of Medicine and HHS, the Community Health Data Initiative is off to a very exciting start (see http://www.hhs.gov/open/datasets/initiative_launch.html). At this event, attended by 400 people in person and 300 people online, innovators demonstrated more than a dozen amazing applications that had been built or significantly improved using HHS data in less than 90 days, in response to an initial challenge issued by HHS in March
- We are setting the goal of at least 100 organizations meaningfully involved in the Community Health Data Initiative by June 2011 – defined as supplying data to the Initiative ecosystem or having built applications as part of the ecosystem
- We are also energetically exploring the potential of Semantic Web to enhance the value of our data and the ability of the public to engage with it. At its heart, the Semantic Web is an innovative extension of standard Web technologies to better deal with data on the Web, by providing a means to give Web addresses to data elements so they can be linked. As the Web of linked documents evolves to include the Web of linked data, we're working to maximize the potential of Semantic Web technologies and realize the promise of Linked Open Government Data at HHS. Since the one year anniversary of the launch of Data.gov this past May 2010, Data.gov is now one of the largest providers of semantic data. HHS is pleased to be leading this community of practice and their collaborative activities inside the Data.gov Program Management Office. In addition, we are working across HHS to best leverage the opportunities that this technology presents to linking HHS open government data across agencies, leveraging our federal government-wide Open Government for Health workgroup and Data Council to catalyze and coordinate these efforts. We believe that data quality and value will improve as more people engage with the data. In light of this, we'll begin to explore social media tools to facilitate the creation of metadata vocabularies and curation of their corresponding instance datasets, with both seen as objects of social collaboration. We think that the 'Social Data Web', where we combine the features and capabilities of the Social Web and the Web of Data, is a powerful idea that will lower our coordination costs and allow independent evolution and interlinking across government information domains while enhancing data quality. There are inspiring examples of socially managed data sites and many existing and emerging tools with strong Semantic Web support to leverage that we intend to explore in the future.
- By employing these processes for ongoing data prioritization, release, and monitoring, HHS intends to increase the value produced by its information resources in several ways. Consumers will be able to access information and benefit directly from using it personally, as when somebody planning to travel familiarizes themselves with disease risks and vaccinations. Beyond that type of use, HHS intends for the information to prove valuable for improving public administration, positively impacting how we and other levels of government can deliver services and accomplish our missions more effectively, more efficiently, more rapidly, and with higher levels of customer satisfaction. A third source of value is the ability for our information to bring new transparency to the health and health care sectors, helping generate insights and spark action to improve performance. A fourth source of benefit is the value of our information to research -- the ability of those discovering and applying scientific knowledge to locate, combine, and share potentially relevant information across disciplines to accelerate progress. Finally, we intend for our massive public information resources to have entrepreneurial value, to serve as a well-maintained community asset available to catalyze the development of new products and services that benefit the public and, in the process of doing so, generate private market economic growth fueled by innovation.
Compliance with IT Dashboard, Data.gov, eRulemaking, Recovery.gov, and USASpending.gov Guidance
HHS is committed to improving access to data posted on central government websites to ensure greater transparency in programs and greater accountability for resources. HHS provides information to the IT Dashboard, eRulemaking, Data.gov, USASpending.gov, and Recovery.gov websites to increase public access to what we do, how we impact needy populations and communities, and how well we are performing in these areas.
The spirit of Open Government affords HHS an exciting opportunity to examine ways to streamline data and systems across functional areas to improve the completeness and the accuracy of our data. This section will describe the Department’s general approach to fostering high quality financial data, and outline its current compliance activities. Additional steps to enhance the quality of financial data and address quality gaps were outlined in HHS’s Open Government Financial Data Quality Plan of June 2010. As addressed in the plan, HHS invites the public to review our current data processes and transparency efforts in order to more fully engage the public in our ongoing efforts to improve data quality and resolve deficiencies in a timely manner.
Federal IT Dashboard
The IT Dashboard is a new website, launched by the Office of Management and Budget in June 2009, which enables federal agencies and the general public to view details of federal information technology investments.
What does the IT Dashboard Do?
The IT Dashboard helps the public review and track the spending, performance, and progress of technology investments over time. Users can obtain more information about the kinds of technology in which HHS is investing, who is performing this work, and other details of technology spending.
How is HHS complying with OMB IT Dashboard Guidance?
HHS has embraced the Federal IT Dashboard transparency imperative and fulfilled all associated requirements by integrating the Federal IT Dashboard with existing HHS IT investment governance processes, assigning HHS Chief Information Officer (CIO) IT investment ratings, focusing on timely updates and data quality, and establishing a new process to address public questions/concerns. The Office of the Chief Information Officer (OCIO) is responsible for administering these activities. HHS actions include the following:
- Integrating Federal IT Dashboard with IT Investment Oversight: HHS has integrated the Federal IT Dashboard into its existing IT review and oversight activities. This includes sending monthly updates of IT investment data to the Federal IT Dashboard and incorporating reviews of information posted on the Federal IT Dashboard as part of monthly HHS IT investment reviews.
- Assigning HHS CIO IT Investment Ratings: In July 2009, in accordance with Federal CIO Rating guidance, HHS OCIO assigned initial CIO IT investment evaluation ratings to all of its major IT investments posted on the Federal IT Dashboard. HHS initial CIO evaluations focused on leading indicators of project success, including performance/risk management, quality of planning, and stakeholder approval.
- Focusing on Timely Updates and Data Quality: Since the Federal IT Dashboard was released, HHS has consistently updated its IT investment information on the Federal IT Dashboard by the last day of the reporting month to ensure the presentation of quality data to the public and for informing IT investment reviews.
- Establishing a New Process to Address Public Questions/Concerns: To engage with the public on IT investments, HHS has established a process for responding to a public inquiry on the IT Dashboard. The HHS OCIO is responsible for coordinating a response to public inquiry with the end communication issued by a Public Affairs representative.
Data.gov
Data.gov is a website sponsored by the Office of Management and Budget and federal agencies to increase public access to high value, “machine readable” federal data sets.
What does Data.gov provide?
Data.gov provides searchable data catalogs that present data in three ways: through a "raw" data catalog, a tool catalog, and a geodata catalog. HHS posts data under the "raw" data catalog and the tool catalog since much of our data are currently available in these formats.
How is HHS complying with the Office of Management and Budget’s Data.gov Guidance?
Since the launch of Data.gov in May 2009, HHS has mobilized its Operating Divisions and programs to identify and submit high-value data sets that allow the public greater access to downloadable data. Please review Section 3.1 for a description of the HHS data currently available for download on Data.gov.
HHS is committed to not only increasing the public’s ability to locate, access, understand, and use the data posted to Data.gov, but is also committed to monitoring and improving the quality of the high-value data sets we release. Details on planned actions to improve the quality of spending information on Data.gov are included in the Department’s Open Government Financial Data Quality Plan.
eRulemaking
eRulemaking is a current government-wide initiative committed to the following objectives:
- Increasing public access to and participation in developing regulations and other related documents that can impact the public
- Promoting more efficient and effective rulemaking through public involvement
How does eRulemaking work?
In 2003, the eRulemaking program launched the Regulations.gov website to enable citizens to search, view and comment on regulations issued by the Federal government.
On average, federal agencies and departments issue nearly 8,000 regulations per year. In the past, if members of the public were interested in commenting on a regulation, they would have to know the sponsoring agency, when it would be published, review it in a reading room, then struggle through a comment process specific to each agency. Today using Regulations.gov, the public can shape rules and regulations that impact their lives conveniently, from anywhere. By accessing Regulations.gov , the public can view and comment on regulations with less burden and more engagement with agencies throughout the eRulemaking process.
How is HHS participating in eRulemaking?
A number of HHS Operating Divisions are complying with eRulemaking goals by using designated eRulemaking systems for their rulemaking activities. The Department’s Food and Drug Administration (FDA) and Centers for Medicaid and Medicare Services (CMS) use the Federal Docket Management System (FDMS) -- a pillar of the eRulemaking initiative -- for their rulemaking business.
Process and Background:
CMS and FDA use FDMS to process all regulations and notices. Specifically, all regulations and notices published in the Federal Register are posted to Regulations.gov. With limited exceptions, public comments are processed and posted at Regulations.gov for public viewing.
- CMS issues an average of 150 Federal Register documents per year. FDA issued 705 Federal Register documents in 2009. The number of comments for each regulation varies, but in 2009 CMS received over 25,000 comments.
Benefits to the Public:
Fewer citizens have to go to FDA in person to view a document. The change has been dramatic. FDA public-room visits from visitors have decreased, from 1,203 in 2007 to 351 in 2009. And as a result of increased web accessibility, related FOIA requests decreased from 1,135 in 2007 to 323 in 2009.
- CMS staff note that FDMS has provided the public with greater access to CMS’s regulations by allowing the public to view the CMS regulations online. In addition, FDMS provides the convenience of allowing the public to submit comments electronically and participate more easily in the rulemaking process.
Benefits to HHS:
- FDMS has allowed better use of staff resources, because the public has virtually stopped submitting paper comments, instead using the FDMS to submit electronic comments
- CMS staff now manages public comments more efficiently. FDMS allows components within CMS to access the comments quickly, which in turn facilitates the development and clearance of policies more quickly
USASpending.gov
USASpending.gov is the Office of Management and Budget’s response to the requirements of the Federal Funding Accountability and Transparency Act of 2006 (Transparency Act or FFATA). The Transparency Act requires a single searchable website, accessible by the public for free that includes for each federal award:
- Name of the entity receiving the award;
- Amount of the award;
- Information on the award including transaction type, funding agency, etc;
- Location of the entity receiving the award;
- Unique identifier of the entity receiving the award.
How does USASpending.gov work?
Data on USASpending.gov are largely obtained from the following sources: the Federal Procurement Data System (FPDS), which contains information about federal contracts; and the Federal Assistance Award Data System (FAADS), which contains information about federal financial assistance such as grants, loans, insurance, and direct subsidies. Data are also obtained from agency submissions via OMB’s FAADS PLUS file format.
How is HHS Complying with USASpending.gov Guidance?
Since 2006 HHS has achieved outstanding performance on USASpending.gov data transmission and compliance. Current performance is based on based on the timeliness and content of data submission. To view current HHS USASpending.gov performance, visit: http://www.usaspending.gov/explore?&carryfilters=on&tab=By%20Agency&fiscal_year=2010&tab=By%20Agency&maj_contracting_agency=75&fromfiscal=yes&overridecook=yes.
What about HHS’ Financial Assistance Data in USASpending.gov?
To send assistance data to USASpending.gov, HHS uses its Tracking Accountability in Government Grants System (TAGGS). Grant data from HHS’ Operating Divisions and Staff Divisions are submitted on a weekly and bi-weekly basis to TAGGS, and these data are reported to USASpending.gov on a bi-weekly basis. HHS has instituted data validation processes to ensure the consistency and accuracy of its grants award data.
What about HHS’ Contract Award Data in USASpending.gov?
HHS uses its Departmental Contracts Information System (DCIS) to collect, report, and transmit contract award data to the Federal Procurement Database System (FPDS). DCIS receives data from HHS’ contract writing systems. HHS relies upon the data validation and edit-check features found within FPDS to ensure the accuracy and completeness of its contract award data. The timeliness of each contracting offices’ completion of the contract action reports is measured on a quarterly basis. These financial data are transmitted to USASpending.gov by FPDS on a bi-weekly basis. OMB posts these data on USASpending.gov on a bi-weekly and monthly basis.
What are the Identified Areas for Improvement?
HHS is building upon its current successes and actively working to improve the quality of financial assistance and contract award data in USASpending.gov. Although HHS has achieved outstanding USASpending.gov performance to date, HHS’ Open Government Financial Data Quality Plan identifies areas for improvement and associated costs, and formalizes a strategy to enhance the quality of spending information and sustain this work overtime.
Recovery.gov
Recovery.gov is the Office of Management and Budget’s website to track and report on American Recovery and Reinvestment Act (ARRA) spending and to report fraud, waste, and abuse.
How is HHS Complying with Recovery.gov Guidance?
HHS has achieved full compliance with Recovery.gov mandates. The Department’s strategic response to the requirements of ARRA and Recovery.gov was an unprecedented effort to mobilize, modify, develop, and enhance staff and resources to oversee the effective execution of over $141 billion in ARRA funding. Specific ARRA activities include the following:
- Submission of ARRA Financial and Activity Reports: HHS submits a weekly ARRA Financial and Activity Report from the Department’s financial management system to the Office of Management and Budget to provide the public with a snapshot of HHS’ Recovery Act obligations and outlays.
- Submission of Bi-Weekly ARRA Transaction Level Data: OMB uses the Recovery Act transaction data sent to USASpending.gov to populate and update the Recovery.gov Total Funding Map, displaying total ARRA funding to States.
- Recipient Reporting Support: HHS provides additional support to ARRA recipients through an innovative web-based tool that assists grantees in locating, tracking, and understanding data for the quarterly recipient reporting requirements. The Recovery Act Recipient Reporting Readiness Tool (RRT), which is available at http://taggs.hhs.gov/ReadinessTool/ enhances data quality for HHS and recipients by ensuring that data are synchronized in recipient and agency systems for quarterly reporting.
As a result of using this tool, HHS achieved a 99.4% OMB recipient reporting success rate. HHS was able to validate that over 99% of ARRA grant recipients complied with FederalReporting.gov requirements. Other achievements include:
- Lowering burden on recipients for reporting;
- Providing recipients a source of information for critical grant award data fields needed for OMB reports; and
- Bridging key gaps between state and federal award data and systems.
As a result of specific Reporting Readiness Tool outcomes, OMB has designated HHS’s Reporting Readiness Tool as the primary model all government agencies should follow to improve ARRA recipient data quality and reporting performance.
How HHS Will Inform the Public About the Business of Our Agency
The Department of Health and Human Services is deeply committed to the principles of Open Government and sees all of our communications efforts as ways to encourage and promote the goals of Open Government. Even prior to the Open Government Directive, HHS was using all of our Public Affairs assets to engage the public and our employees.
From the web videos, webcast forums and weekly reports on healthreform.gov to the flu symptom tool and the flu vaccine finder on flu.gov, to the helpful safe food tips on foodsafety.gov to the fraud finder map on stopmedicarefraud.gov, our goal has been to centralize and focus our communications efforts across HHS into easy one-step consumer campaigns.
We learned many important lessons during the 2009 H1N1 flu outbreak that we will be applying to our Open Government efforts. As part of our communications efforts around this pandemic, we developed new tools and protocols for the web, for our studio and for our advertising and media efforts that are not only helping us in Open Government efforts, but are also helping to change the way we have done business in the past. We will be building upon those principles, policies and practices as part of HHS’s commitment to Open Government.
Our senior leadership will play a critical role in our efforts to communicate to the public about the transparency, collaboration and participation improvements we are making here at HHS. Secretary Sebelius is a strong supporter of New Media and from her first day in office has recorded web videos, done Facebook chats and led our webcasts.
3.5.1 Accessibility
Open Government starts with ensuring that every American has access to the life saving health and human services materials and programs that HHS provides. HHS believes that “open” means open to all; we will do our utmost to remove barriers to participation by persons with disabilities. HHS is acknowledged as a leader in providing access, and freely shares its 508 standards, training, testing facilities, knowledge and experience and will continue to work at making its website information accessible to all.
3.5.2 Assets
Broadcast
HHS and our operating divisions have several full-service television studios with experienced staff that can create and produce public service announcements, public webcasts, full-scale TV shows, daily press tours and press conferences. We are also investing in mobile video technology that can be used for web videos, interviews and press events. Wherever possible, we use our strong web and broadcast assets to promote training, research and collaboration.
Web
The department’s websites, totaling more than 100 million pages, contain vast amounts of information. For legacy reasons, web content is largely organized by originating office or program. It’s like a library whose bookshelves are organized by publisher. Utilizing content syndication and other emerging technologies, as now practiced on www.flu.gov, HHS is working to overlay a topically organized web experience to satisfy consumers’ need for one-stop access to primary information.
Our work in food safety, AIDS, and flu tell us that the public wants to see our information organized in a way that presents clear, easy access. Later this year we will create new tobacco prevention and vaccine sites that will be modeled on the one-stop principle for presenting information.
We use usability testing and site visitor feedback to design our web pages and communications materials so that people can find what they want, when they want it. We strive to present consumer information using plain language.
New Media Center
The department has established a New Media Center to promote and coordinate the growing use of New or Social Media across the department. Social Media fosters direct interaction between the public and the department; enlists the public in the viral dissemination of important information; makes departmental health information available on countless other websites and applications; facilitates the use of open-source data sets and engages new demographic audiences in the public discourse. Social Media has already been used to convey critical health safety information addressing concerns ranging from the H1N1 flu pandemic to food safety recalls.
3.5.3 Our Plan
Over the past year, HHS has adopted a specific set of policies and continues to develop others to increase our Open Government communications efforts. We have also initiated a series of activities to further improve Open Government at HHS. They are detailed below. Some of these are quick fixes and others will require work across the department.
Increased Access to HHS Events & Information
Beginning in April, HHS will publish a schedule of key events occurring across the Department each week, including events featuring key Departmental officials.
Whenever possible, we are working to webcast and tweet HHS press conferences, media events and informational briefings. Secretarial and Deputy Secretarial public/press events are taped and transcripts are made available on the web. We are working to add audio and video transcripts to our library on the website.
To bring transparency to one of our primary forums for public participation, starting in April, the Open Government website will link to all of the Department’s Federal Advisory Committees.
Webcasts & Videos
HHS frequently uses webcasts and on-line videos to highlight the department’s priorities, and we will be working to expand this capacity throughout the coming year.
We are working to create a library of on-line videos/tutorials to help consumers find critical data and information. Videos will show viewers how to use the tools on www.hhs.gov/open and our other one-stop websites and find the resources they need across the department.
One idea we are pursuing is to work with our Operating Divisions to create a series of day-in-the-life Web vignettes about individuals across the department, in which they describe how their jobs help Americans get critical health and human services.
In late spring 2010, we are planning to unveil a weekly “Ask the Secretary” video feature on www.hhs.gov/open where the Secretary will answer a few questions each week that exemplify the range and types of questions that we are receiving from the public via the mail, the phones and on our websites.
Starting in May, the Department plans to schedule monthly webcasts on departmental priorities (a 30 minute show during the lunch hour broadcast live on hhs.gov). The webcasts will be hosted by the Secretary, Deputy Secretary and departmental leadership to discuss the program priority and the leaders behind it. News announcements and a featured 10 minute question and answer period from your emails and phone calls will often be featured on the webcasts.
Training
We will hold twice yearly Open Government trainings department-wide for communications staff. Training will also be provided for FOIA officers and coordinators.
HHS Open Government Website
The HHS Open Government website (www.hhs.gov/open) is first and foremost is a place where people come to work. It is designed to foster and model transparency. Content will value actions over words. Evaluation of and reaction to public input will be a hallmark of this site. The website fulfills three fundamental purposes:
- To facilitate the public’s ability to access, interface and interact with the department’s policies, goals and business processes.
- To make the department’s informational holdings accessible in open and logical formats that permits their fullest utilization.
- To present the department’s consolidated Open Government Plan in a transparent, user-friendly venue that invites broad public engagement.
Beginning with the release of our Open Government Plan Outline, each plan iteration will be presented on the Open Government website for public comment and discussion. The public is also asked to recommend data sets, tools, participation opportunities and other information that could be made openly available.
In addition, HHS Chief Technology Officer Todd Park blogs weekly on key Open Government Plan elements, suggests areas for discussion, and responds to plan comments and recommendations. To further the dialogue, his blog also invites general comment and discussion.
The Department’s senior leadership will also post updates each on the Open Government Blog related to who we are and what we do.
Information and Data Sharing
HHS is committed to presenting its massive collections of data in accessible formats that allow and encourage the fullest use of this data. These open-format data collections will be made available on the Open Government website via a searchable contextual database. The site will likewise provide contextual database access to the department’s many tools that facilitate transparency. HHS is committed to building on all of these collections and to listening to you regarding not only what is there, but also the best way to present our vast holdings.
The Open Government website will present the department’s Annual Freedom of Information Act (FOIA) report in multiple accessible formats. As we discuss in detail later in the plan, HHS is committed to improved implementation of FOIA as we proceed with our Open Government plans. By significantly increasing web-based access to information and documents of interest, the Department is committed to reducing the need for people to submit FOIA requests.
HHS is making increasing use of new or social media to interact and interface with the public. The Open Government website provides direct one-stop access to the Department’s new media engagement opportunities, including those on Facebook, Twitter and YouTube.
Privacy Splash Page – Spring 2010
The companion of Open Government is a government that protects and advocates privacy – especially the privacy of people’s personal medical information. To facilitate this, we plan to create a “privacy home page” that helps the public easily access information about the array of privacy protection programs that exist, the new regulations that are being created and the key privacy personnel who work every day behalf of the American people.
Trusting that your privacy will be protected as part of the on-line experience across the department is critical. It is important that HHS be an IT model of how electronic medical records and other new innovations are protective of privacy information while achieving the benefits of making information easily accessible on-line.
How HHS Is Meeting Current Records Management Requirements
The results of the Records Management program at HHS are published on a publicly available website: http://www.hhs.gov/ocio/policy/recordsmanagement/index.html. These results demonstrate how HHS is carrying out records management requirements in accordance with NARA regulations and OMB mandates.
HHS and Congressional Requests for Information
The legislative branch of our Federal government has intense interest in the work conducted by the Department of Health and Human Services. Like the public, Members of Congress and their staff frequently direct letters, emails, and phone calls to the Department that require timely and responsive reply.
Responding to Congressional Inquiries: Workflow Practices
The topic areas of interest to Congressional Members are very broad in range as is the nature of the requested action. For example, some requests may be on behalf of a Member’s constituent who is looking for certain types of information or assistance with an issue. In other cases, Members request formal reports, technical documents, budget information, and answers to key questions regarding a particular issue.
The Office of the Assistant Secretary for Legislation (ASL) coordinates Congressional requests across the Department. Members submitting requests may come directly to ASL or to agency legislative affairs offices. ASL performs many additional functions, including developing responses to requests on behalf of the Secretary and staff offices within the Office of the Secretary. ASL also coordinates testimony, clearance of proposed legislation and responses to inquiries about them.
With regard to formal incoming requests, to coordinate the response to a request from a Member, ASL assigns a lead office/agency and an ASL staff member to coordinate development of the response and clearance of the transmittal to the Member’s office. For these situations, the assignment often requires a “delivery date” for formulating the draft response. In other cases, Members or their staff may make informal inquiries to ASL that may be handled less formally through phone conversations or email.
Proactively, ASL may also contact Members’ offices for those who are very interested in particular topics to inform them of upcoming events, reports, or activities that may be of interest. Throughout the process, ASL and other legislative offices maintain working knowledge of legislative activities of relevance to health and human service activities. ASL and HHS agency legislative offices track pending issues closely on proposed legislation, arrange for responses to requests, support expert testimony, and coordination of clearance for specific issues across the government.
ASL Divisions
The office consists of six divisions:
Immediate Office of the Assistant Secretary for Legislation: Serves as principal advisor to the Secretary with respect to all aspects of the Department's legislative agenda and Congressional liaison activities.
Office of the Deputy Assistant Secretary for Discretionary Health Programs: Works on the legislative agenda and serves as the lead liaison for discretionary health programs. This portfolio includes health-science-oriented operating divisions of the department including the National Institutes of Health, the Food and Drug Administration, the Centers for Disease Control and Prevention, the Health Resources and Services Administration, the Office of the Assistant Secretary for Preparedness and Response, and the Surgeon General, among others.
Office of the Deputy Assistant Secretary for Mandatory Health Programs: Works on the legislative agenda and serves as the lead liaison for the Centers for Medicare and Medicaid Services as well as the Indian Health Service.
Office of the Deputy Assistant Secretary for Human Services: Works on the legislative agenda and serves as the lead liaison for the departments’ human services and aging program divisions.
Congressional Liaison Office: Serves as the lead liaison to Members of Congress by notifying them of departmental activities and initiatives, maintaining the Department's grant notification system and coordinating agency response to congressional inquiries.
Office of Oversight and Investigations: Responsible for all matters related to Congressional oversight and investigations, including those performed by the Government Accountability Office.
The organizational structure chart and staff list for the Office of the Assistant Secretary for Legislation can be found at: http://www.hhs.gov/asl/divisions/divisions.html#clo
Grants
The ASL Congressional Liaison Office (CLO) responds to congressional inquiries about grant awards; notifies congressional offices of grant awards made by the Department; and facilitates technical assistance regarding grants to Members of Congress and their staff.
ASL grant information can be found at: http://www.hhs.gov/asl/Grants/grants.html
Testimony
A complete listing of testimony by the Secretary and other Department officials before the United States Congress.
HHS testimony can be found at: http://www.hhs.gov/asl/testify/2010/testimony.html
3.9 – HHS and Information Declassification
In general, most documents held at HHS that have a national security classification were originally classified by another department or agency. Decisions and the process for the declassification of this material rest with their originators.
HHS does have original classification authority and has classified a small number of documents. Declassification of documents, due to time or lack of continuing need for protection, is executed via specific and routine review.
Declassification Authority
The authority to declassify information rests in the following officials:
- The Secretary with respect to all information over which HHS exercises final classification authority;
- The original classification authority, as designated by the Secretary, a successor of the original classification authority, or a supervisor of either;
- The official of the originating agency who authorized the original classification;
- The Director, Office of Security and Strategic Information (OSSI), with respect to all classified documents originated by a HHS-predecessor agency and being retained for some official reason, following the coordination with the HHS operating division or staff division that has subject matter interest in the documents.
When there is some doubt concerning the classification of a document, the information must be transmitted for review to the Director, OSSI, for review and to an agency with proper subject matter interest and original classification authority -- at which point that the agency will decide to declassify, or extend the initial classification level.
Annual Review
All classified documents in the possession or control of an organization are subject to an annual review conducted by the Classification Security Officer (CSO) of the organization. This review is conducted to identify documents that require declassification or destruction and must be accomplished prior to the HHS Annual Status Report on Classified National Security Information.
Automatic Declassification
All classified documents will have a maximum classification life of 25 years from the date of its original classification, unless the Director of the Information Security Oversight Office within the National Archives and Records Administration has determined that the document may be exempt from automatic declassification.
Mandatory Review Requests for Declassification
Anyone may request a review for declassification of information. These requests are submitted to the Director, OSSI, as either a mandatory review request or under the Freedom of Information Act (FOIA) review process.
If the request is approved, the Director, OSSI, must then declassify all HHS-originating information by marking it to reflect the change, authority for and date of declassification. If the requested information cannot be declassified in its entirety, declassified portions that constitute a coherent segment are released, and if the information cannot be released in whole or in part, the action office must provide the reasons for denial. In cases where declassification is denied, in whole or in part, the Director, OSSI, in coordination with the HHS Freedom of Information Act office, must notify the requestor of the final determination and reasons for denial, as well as the right to appeal within 60 working days of the receipt of the denial.
HHS may also require a fee for declassification review requests, which may be appealed if the requested information is not declassified and released in whole.
HHS and the Freedom of Information Act (FOIA)
3.7.1 Overview
The Freedom of Information Act (FOIA) gives the public the right to request records held by the federal government. Many agencies hold large backlogs of requests. The Open Government Directive recognizes this, and calls for a 10% annual reduction in “significant” backlogs. The HHS annual backlog at the end of September 2009 was over 17,000 requests.
To address this backlog and as a commitment to the Open Government Directive, HHS has initiated a strategic sixteen-week HHS Open Government FOIA Project with a dedicated project manager and team. This project is to assess the health of HHS FOIA operations, identify and prioritize improvement opportunities, and define a roadmap to implement the improvements.
The project is currently in progress. The project scope encompasses review of FOIA operations within the Office of the Secretary; the Program Support Center (PSC); and eleven HHS Operating Divisions, including the Centers for Medicare and Medicaid Services (CMS), Food and Drug Administration (FDA), National Institutes of Health (NIH), and Centers for Disease Control and Prevention (CDC). In addition, it includes collaborating with the FOIA offices of other federal agencies, to learn from successes in addressing FOIA backlogs. Some of the preliminary findings from the assessment, and the plan thereof, are presented in the subsequent sub-sections. Once the project is complete, this section of the HHS Open Government Plan will be updated with further details.
Section 3.7.2 provides a high-level FOIA assessment. Section 3.7.3 provides an analysis and preliminary plan, also at a high level.
There is no single improvement that will solve the majority of the challenges facing HHS when it comes to FOIA. In the spirit of openness, many of the ideas in this plan are being shared while still under early investigation. However, it is clear that a multi-pronged approach will be necessary for backlog reduction: enhanced FOIA program efforts, the implementation of metrics for FOIA office performance, and an increased use of supporting technologies.
3.7.2 Assessment: Current State
3.7.2.1 Organization
- FOIA Organization at HHS is Decentralized. There are thirteen HHS FOIA offices, located in various Operating Divisions and Staff Divisions (components of the Office of the Secretary). Each FOIA Office is run by a local FOIA Officer. Operating Divisions such as FDA and NIH have numerous “satellite” FOIA offices located within their institutes and centers. The entire FOIA program, though decentralized, is the responsibility of the HHS Chief FOIA Officer, the Assistant Secretary for Public Affairs.
FOIA offices and related positions (e.g., the FOIA public liaisons) are managed by the leadership of their respective Operating Divisions and Staff Divisions, including budgeting and staffing decisions
- Funding Towards FOIA is not Transparent to the FOIA Offices. Most FOIA offices do not have their own budgetary allocation. Instead, their funding comes from the offices to which they report. The reporting structure of FOIA offices within different Operating Divisions varies. For example, in the FDA, the FOIA office is part of the Office of Public Information and Library Services, whereas at CMS it is part of the Office of Strategic Operations and Regulatory Affairs.
- 88% of FOIA Requests are Concentrated in Two Agencies. The number of FOIA requests received varies greatly across the Department. For Fiscal Year 2009 (FY09), the Department as a whole reported receiving 48,564 requests. Of these, 88% were received by two FOIA offices – 67% (32,541) went to the CMS FOIA office, and 21% (10,337) went to the FDA FOIA office. The remaining 12% of requests were received by the other eleven FOIA offices. Of these, the CDC and NIH FOIA offices received 3% (1272 and 1270, respectively) each. The Administration on Aging (AOA) received the least number of requests (only ten).
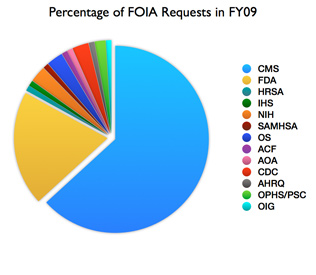
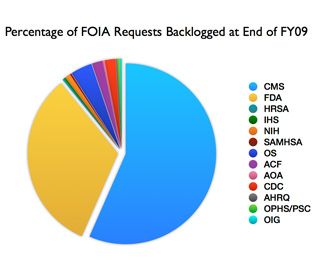 93% of Backlogs are Concentrated in Two Agencies. Within HHS, 59% of backlogged requests (10,312 requests) are with CMS, and 34% (4,818) are at FDA. The next largest contributor is the Office of the Secretary (OS) FOIA Office, which holds only 4% of the backlogged requests. The Department is therefore focusing on these areas for backlog reduction.
93% of Backlogs are Concentrated in Two Agencies. Within HHS, 59% of backlogged requests (10,312 requests) are with CMS, and 34% (4,818) are at FDA. The next largest contributor is the Office of the Secretary (OS) FOIA Office, which holds only 4% of the backlogged requests. The Department is therefore focusing on these areas for backlog reduction.
Doing More With Less
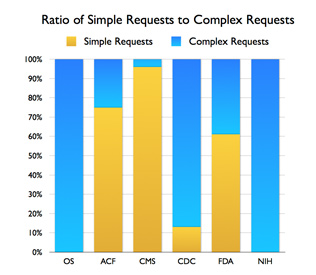 FOIA Requests Vary in Complexity. Not all requests are equivalent. Some requests require the careful review of thousands of pages, while other requests are easily granted with a single page record that needs no redaction. The definitions for “simple request” and “complex request” vary between FOIA offices, as do the definitions for “partially fulfilled” and “fully granted.”
FOIA Requests Vary in Complexity. Not all requests are equivalent. Some requests require the careful review of thousands of pages, while other requests are easily granted with a single page record that needs no redaction. The definitions for “simple request” and “complex request” vary between FOIA offices, as do the definitions for “partially fulfilled” and “fully granted.”
- Backlog reduction efforts are decentralized. Under the Open Government Act of 2007 and Executive Order 13392 (2005), agencies were directed to develop FOIA improvement plans with backlog reduction goals. At HHS, no comprehensive or department-level activities were implemented in this regard. However, overall department backlogs were lowered as some Operating Divisions have shown progress individually in the last few years.
3.7.2.2 People
- FOIA Analysis Requires Specialized Knowledge. . The responsibilities of FOIA staff vary greatly, and only some perform the disclosure analysis necessary before a record can be released. FOIA disclosure analysis requires knowledge of not only the Freedom of Information Act and the amendments, executive orders, and Operating Division policies that have augmented it, but also comprehension of the 36 years of case law that have come since the Act itself and related statutes as well. In addition, some HHS Operating Divisions, such as FDA, review documents that are rich in technical and scientific content – work which requires additional educational background, knowledge and training to process and accurately redact requests.
- Operating Division FOIA Operations Vary in Scale. There are 264 full-time equivalent (FTE) FOIA personnel at HHS. Of the thirteen HHS FOIA offices, all have less than seven of these FTEs except NIH (30 FTEs), CMS (67 FTEs), and FDA (124 FTEs, up recently from 108). Almost all of the FTEs at these three Operating Divisions are satellite personnel, with different roles depending on the Operating Division.
3.7.2.3 Process
- FOIA Requests Mostly Arrive by Mail. Most HHS FOIA offices do not offer an online FOIA request form, so requests come largely by mail, fax, email, and (for commercial requestors) courier services. The Departmental FOIA office offers a web form that generates an email to the Office of the Secretary’s FOIA office, which then manually forwards the email to the appropriate HHS FOIA office. While this adds convenience for the requester, no internal efficiencies are gained through the current system.
- Request Tracking Capabilities are Minimal. All Operating Divisions are required to provide tracking information to their requestors, but only CDC allows users to do this on the web; the rest handling status tracking more manually. Requests are processed internally at the smaller FOIA offices, but the larger operations at CMS and FDA have most of their requests fulfilled by satellite FOIA personnel.
- Annual Reporting is Manual. Although the annual report requires the collection of several numbers that might serve as good performance indicators, this is undermined by inconsistencies in the definitions for the terms in the report. A complex request in one FOIA office might require a significantly different amount of work than a complex request in another. The annual report shows nothing of whether long delays are due to fundamentally difficult redaction questions, due to slow response time of the program holding the records, or due to a backlog in disclosure analysis. Since the Department’s annual report is compiled manually, more frequent collection of metrics is not feasible without systematic changes.
3.7.2.4 Technology
- Technology is Underutilized. The challenges encountered in compiling the annual report underlie the differences in technology utilization across the HHS FOIA offices. Most of the HHS FOIA offices do not offer online submission of requests. There are little or no common standards connecting any of the systems supporting HHS’s FOIA offices.
- Technology at CMS. CMS currently uses Strategic Work Information Folder Transfer (SWIFT) to manage its correspondence and is in the process of adding more FOIA-specific functionality to this software. CMS’s backlogged requests, however, are not yet part of this new system.
- Technology at FDA. FDA as a whole is using an in-house customization of Documentum to track Operating Division tasks and dockets, including the tracking of FOIA requests. FDA’s FOIA office is beginning to use commercial software for redaction, and most component offices have used redaction software for years.
- Technology at NIH. NIH uses a custom-built tracking system, and uses physical redaction tape or commercial software for redaction depending on the NIH program office.
- Technology in OS. The Office of the Secretary’s FOIA Office uses Microsoft Access to log requests, and prepares the annual report in Microsoft Word.
3.7.3 Analysis and Preliminary High-Level Plan
The following proposals are high-level ideas, presented to receive public and employee input. Amendments will be made based on input or feedback received.
3.7.3.1 Better Use of Available Information
 Trends and Outliers in Requests. FOIA requests are one of the most reliable and quantifiable metrics for the public’s interest in government. However, throughout HHS, FOIA is very rarely part of the strategic planning process. FOIA should inform Open Government activities, the proactive publishing of datasets, and website content. Accordingly, HHS will look into integrating the FOIA offices into Operating Division and departmental processes, committees, and meetings (e.g., capital planning, the Data Council, the CIO Council, the Innovation Council, the Web Council, budget and contract planning, strategic planning, Open Government and transparency, etc.).
Trends and Outliers in Requests. FOIA requests are one of the most reliable and quantifiable metrics for the public’s interest in government. However, throughout HHS, FOIA is very rarely part of the strategic planning process. FOIA should inform Open Government activities, the proactive publishing of datasets, and website content. Accordingly, HHS will look into integrating the FOIA offices into Operating Division and departmental processes, committees, and meetings (e.g., capital planning, the Data Council, the CIO Council, the Innovation Council, the Web Council, budget and contract planning, strategic planning, Open Government and transparency, etc.).  Prioritizing Information for Proactive Release. The FOIA workload can be reduced, and service to the public can be improved, by the proactive publishing of frequently requested datasets. HHS will establish governance to include FOIA officers across the department in transparency and data sharing planning activities.
Prioritizing Information for Proactive Release. The FOIA workload can be reduced, and service to the public can be improved, by the proactive publishing of frequently requested datasets. HHS will establish governance to include FOIA officers across the department in transparency and data sharing planning activities. - Submission and Tracking of FOIA Requests. Requestors are already able to ask FOIA offices for the status of their request, but a robust tracking system would make this available online and could include the status of all requests. HHS will explore setting minimum standards for the online submission and tracking of FOIA requests across all Operating Divisions. Tracking requests is also crucial to ensuring appropriate resources are allocated for FOIA operations; this is discussed in 3.7.3.2.e, below.
- Technology Architecture. HHS is actively investigating whether the centralization or federation of technological resources used in processing FOIA would be a significant improvement over the current decentralized approach. Similarly, the HHS FOIA websites vary in features and content, and HHS is investigating whether more uniform standards or centralization of HHS FOIA web tools would bring added value to the public.
- Instituting a Performance Management Culture. Metrics showing the health of the FOIA program will be defined, and systems will be put in place to capture and report metrics to the Chief FOIA Officer on a least a monthly basis. FOIA metrics developed by HHS will be shared with other agencies.
3.7.3.2 Ideas for Improving FOIA Processes
The following list is preliminary and presented in order to receive input on ways to strengthen FOIA at HHS, address the deficiencies that have led to the significant backlog of requests, and achieve excellence in FOIA administration. This list is not a final plan; the assessment of HHS FOIA operations and planning for remediating issues is still in progress.
- Communicating FOIA Needs. The decentralized FOIA operations at HHS allow FOIA offices to be close to the programs that have the information that is being requested. It is possible to improve communication between FOIA offices and Operating Division leaders through the same strategy described in 3.7.3.1.a; that is, better integration of FOIA into existing activities.
 FOIA as a Senior Executive Priority. While FOIA needs will be better addressed through increased participation of FOIA personnel in Operating Division governance, leadership attention to FOIA performance will also make a difference. HHS will explore methods for increasing senior management attention to improving FOIA operations within its operating divisions.
FOIA as a Senior Executive Priority. While FOIA needs will be better addressed through increased participation of FOIA personnel in Operating Division governance, leadership attention to FOIA performance will also make a difference. HHS will explore methods for increasing senior management attention to improving FOIA operations within its operating divisions. FOIA as a Program Officer Priority. Federal employees who are not FOIA staff play a crucial role in fulfilling FOIA requests. Such individuals hold the records sought through the FOIA process, and are a critical step in the process. However, communication to employees about FOIA responsibilities and statutes is lacking. In addition, few receive training in FOIA. HHS plans to consider ways in which FOIA compliance and training as can be promoted and measured as part of an HHS-wide effort.
FOIA as a Program Officer Priority. Federal employees who are not FOIA staff play a crucial role in fulfilling FOIA requests. Such individuals hold the records sought through the FOIA process, and are a critical step in the process. However, communication to employees about FOIA responsibilities and statutes is lacking. In addition, few receive training in FOIA. HHS plans to consider ways in which FOIA compliance and training as can be promoted and measured as part of an HHS-wide effort.- Matching Skills and Needs. There is no FOIA-specific job series within the federal government, which means there is no government-wide standard for the skills and knowledge necessary to perform FOIA work, no standard for evaluating performance by FOIA staff, and considerable inconsistency in how duties map to salary grades. This need can be met, at least in part, through the efforts to create HHS standards, by better capturing metrics, and through better tracking of requests.
- CMS FOIA Backlog Planning. CMS has developed a short term strategy to take necessary steps to increase case production of existing staff, align contractor resources, and focus regional resources to decrease the existing backlogs. CMS has been charged with developing a long range plan to ensure new backlogs are not created; this plan will include an analysis of resources that may be needed moving forward as well as addressing the need for all aged, pending complex cases to be converted from paper to electronic files for better case management and electronic review and redaction.
- FDA FOIA Backlog Planning. At FDA, of 4,600 backlogged requests, the Center for Drug Evaluation and Research (CDER) and Center for Devices and Radiological Health (CDRH) have approximately 2,000 each. Both offices are built around FOIA staff with not only the standard amount of FOIA knowledge, but also a familiarity with the underlying technologies/sciences specific to their program areas. FDA is committed to backlog reduction through a multi-pronged approach. The agency is currently evaluating the best alternatives to achieve this goal, and anticipates building on the significant (75%) backlog reduction that has occurred since FY2007. Much of the FDA reduction was accomplished by categorizing and resolving large sets of requests in the backlog, and with these requests now closed, the remaining backlog may require new approaches.
 Office of the Secretary FOIA Office Backlog Planning and Program Development. The Office of the Secretary is doing everything it can to reduce its backlog, and is considering additional FOIA-specific activities, including maintaining department FOIA regulations, creating a department-wide FOIA council, and providing cross-training for both FOIA and non-FOIA staff. Also being considered are mechanisms to cooperate with the new Office of Government Information Services’ FOIA mediation efforts and regular meetings between FOIA professionals and the agency Chief FOIA Officer.
Office of the Secretary FOIA Office Backlog Planning and Program Development. The Office of the Secretary is doing everything it can to reduce its backlog, and is considering additional FOIA-specific activities, including maintaining department FOIA regulations, creating a department-wide FOIA council, and providing cross-training for both FOIA and non-FOIA staff. Also being considered are mechanisms to cooperate with the new Office of Government Information Services’ FOIA mediation efforts and regular meetings between FOIA professionals and the agency Chief FOIA Officer.
3.7.3.3. Doing More With Less
 Communication for Greater Efficiency. The simplest method for reducing FOIA backlog and workload is to encourage FOIA staff to work with the requestors to make sure their requests are being interpreted as intended. This method is proven in some components of HHS but is not routine across the department. To reduce the routine waste in fulfilling too broad, obsolete, or ambiguously worded requests, HHS may institute policy regarding communication between the analyst and requestors and establish guidelines to improve direct communication between reviewers and requestors.
Communication for Greater Efficiency. The simplest method for reducing FOIA backlog and workload is to encourage FOIA staff to work with the requestors to make sure their requests are being interpreted as intended. This method is proven in some components of HHS but is not routine across the department. To reduce the routine waste in fulfilling too broad, obsolete, or ambiguously worded requests, HHS may institute policy regarding communication between the analyst and requestors and establish guidelines to improve direct communication between reviewers and requestors.- Technology. As explored in earlier sections, technology solutions can improve FOIA service to the public and management oversight, as well as bring new levels of transparency to the process itself. Technology investments should also result in faster responses and more efficient disclosure analysis; HHS will include these priorities in considering new technology to support FOIA operations.
3.7.4. Conclusion
These are high-level preliminary plans, and will be updated based on public and employee feedback. Public input will help ensure that the agency meets its goal of excellence in FOIA administration.
More background on the department’s FOIA program is available in the 2009 annual FOIA report and the Chief FOIA Officer’s report, both of which are available on the HHS FOIA website.
HHS and Information Declassification
In general, any documents held at HHS that have a national security classification bear that classification from another source. Decisions and the process for the declassification of this material rest with their originators.







